IMpact-RSV Study
SYNAGIS significantly reduced RSV-related hospitalisations by more than half1,2
Preterm infants ≤35 wGA and ≤6 months of age, and children ≤2 years of age with BPD requiring treatment1,2
SYNAGIS significantly reduced RSV-related hospitalisations by more than half
SYNAGIS significantly reduced hospitalisations associated with RSV disease by 39%1,2
Subgroup Analysis
Children ≤2 years of age with BPD requiring treatment1,2
SYNAGIS significantly reduced hospitalisations associated with RSV disease by 78%1,2
Subgroup Analysis
Premature infants ≤35 wGA and ≤6 months of age without BPD1,2
SYNAGIS significantly reduced total days of RSV-related hospitalisation and total days with increased supplemental oxygen2
Premature infants ≤35 wGA and ≤6 months of age, and children ≤2 years of age with BPD requiring treatment2
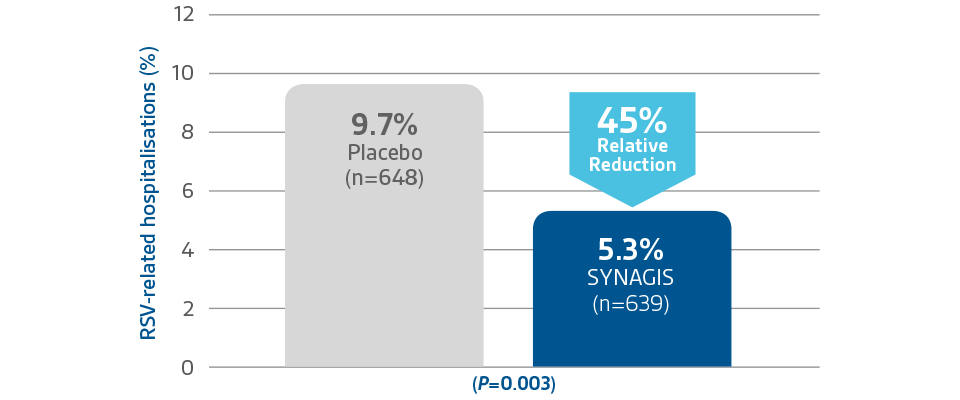
CHD-1 Study
SYNAGIS significantly reduced RSV -related hospitalisations by 45%1,3
Children≤ 2 years of age with HS-CHD1,3
SYNAGIS reduced length of RSV-related hospitalisations by 56% and days of increased supplemental oxygen by 73%1,3
Children ≤2 years of age with HSCHD1,3
Length of hospital stay
Increased supplemental oxygen
SAFETY PROFILE
Safety profile well established in over 20 years of clinical use1,4
ADVERSE EVENTS TABLE:
* Frequency of Anaphylaxis is unknown and frequency for acute hypersensitivity reactions is very common (1/10).
+Identified from post-marketing surveillance.
CONTRAINDICATIONS:
SYNAGIS is contraindicated in children who have had a previous significant hypersensitivity reaction to the active substance or to any of the excipients listed in Section 6.1 of the SmPC, or to other humanized antibodies1.
For full safety and prescribing information, please see the SYNAGIS Summary of Product Characteristics (SmPC).
BPD = bronchopulmonary dysplasia; CHD = congenital heart disease; HS-CHD = hemodynamically significant-congenital heart disease; KSA = Kingdom of Saudi Arabia; IM = intra-muscular; MedRA = medical dictionary for regulatory activities RSV = respiratory syncytial virus; wGA = weeks gestational age;
References: 1.SYNAGIS(palivizumab). Summary of Product Characteristics. 2.The IMpact-RSV Study Group. 1998;102(3):531-537. 3.Feltes TF, et al. J Pediatr. 2003;143(4):532-54. 4.Resch B. Hum Vaccin Immunother. 2017;13(9):2138-2149.

.png)