Safety Data
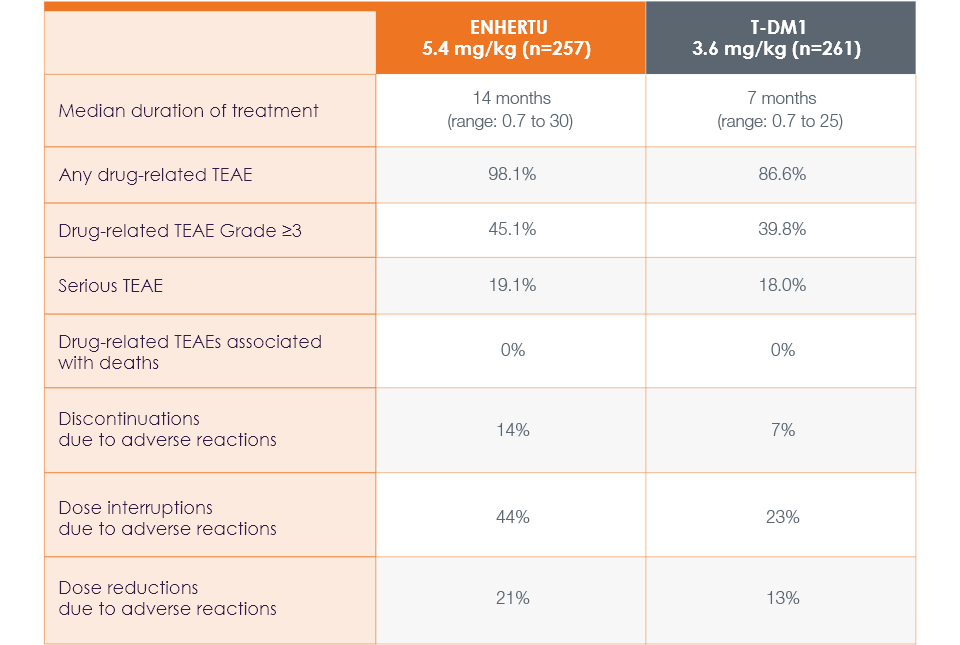
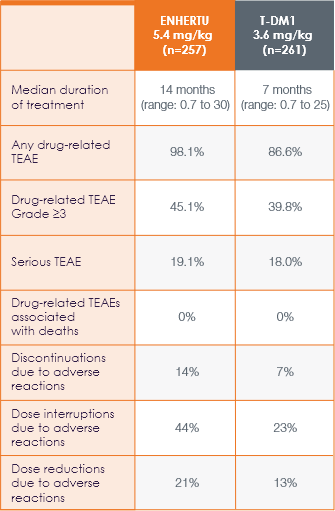
Safety data from DESTINY-Breast03 confirmed the benefit-risk profile of
ENHERTU for HER2+ mBC (5.4 mg/kg) demonstrated in prior studies1,a
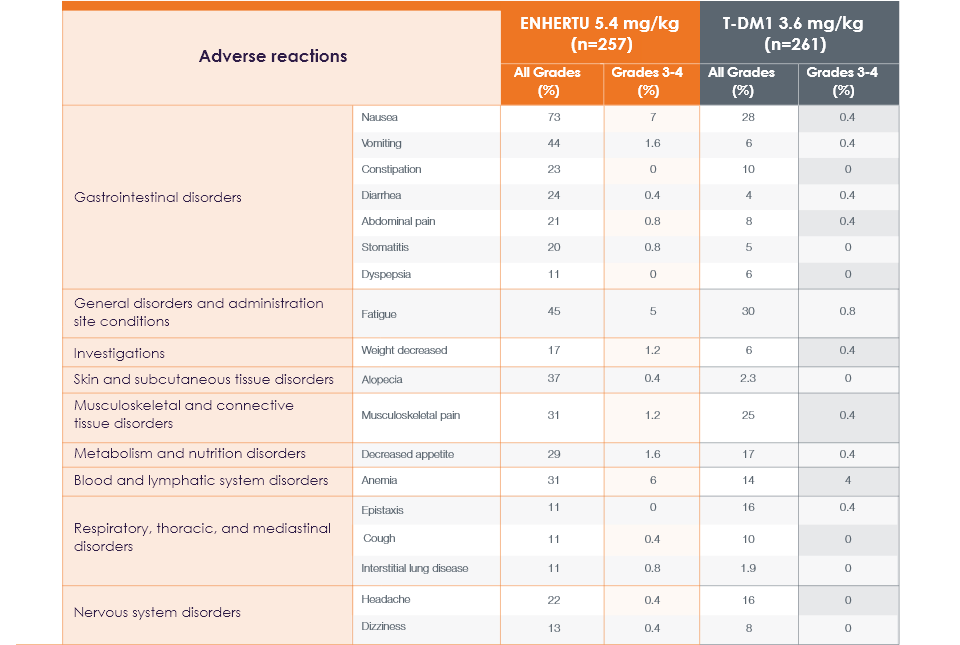
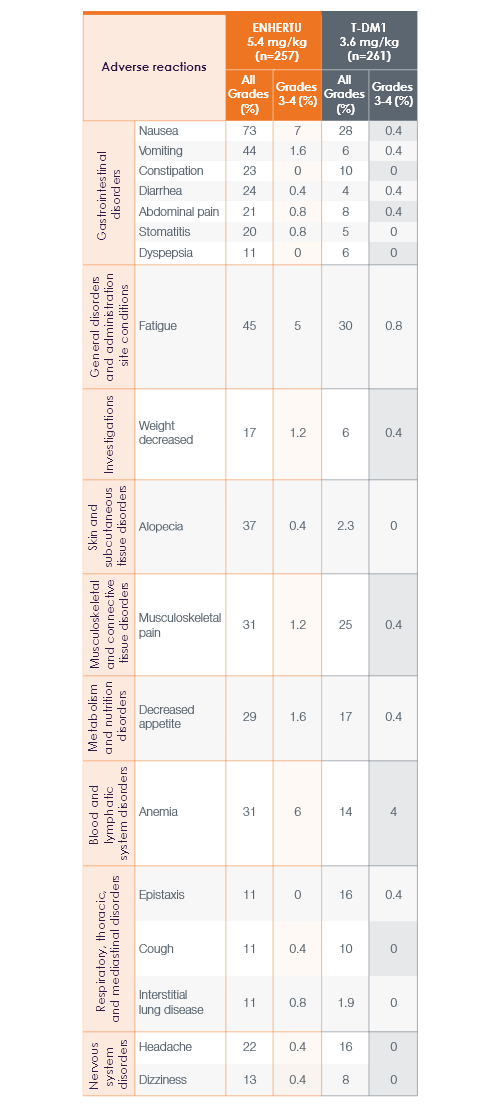
The majority of adverse reactions were Grade 1 or 2
Median duration of treatment was 14 months (range: 0.7 to 30) with ENHERTU and 7 months (range: 0.7 to 25) with T-DM11
Primary endpoint demonstrated superiority in progression-free survival as assessed by BICR
Common adverse reactions (≥10% All Grades or ≥2% Grades 3-4) in patients in DESTINY-Breast031,2
aPrior ENHERTU HER2+ mBC studies: Phase 2 DESTINY-Breast01 and Phase 1 DS8201-A-J101.3
In 2L HER2+ mBC, DESTINY-Breast03 confirmed the positive benefit-risk profile of ENHERTU (5.4 mg/kg, n=257) demonstrated in prior 3L HER2+ studies1,4
Management of select common adverse reactions
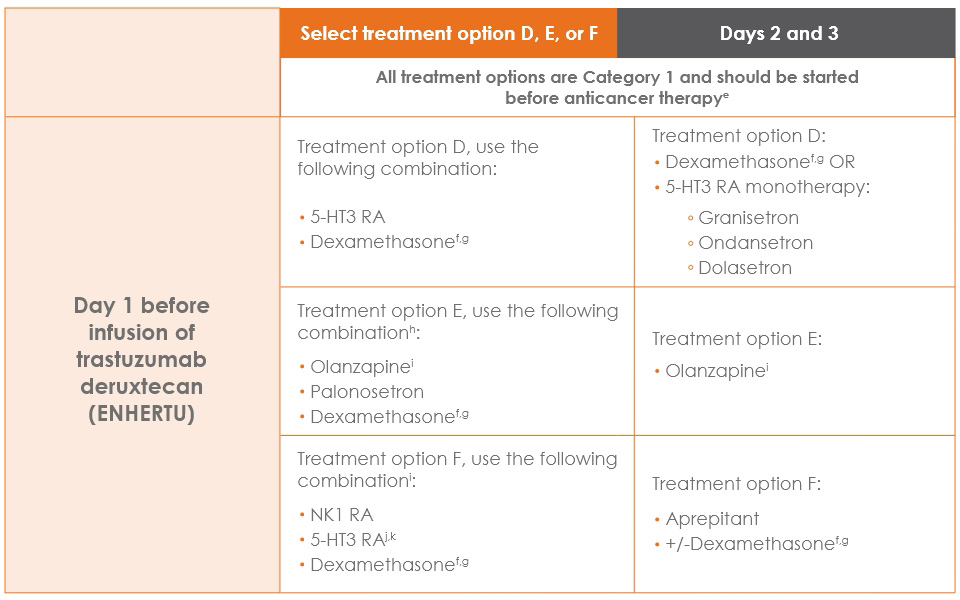
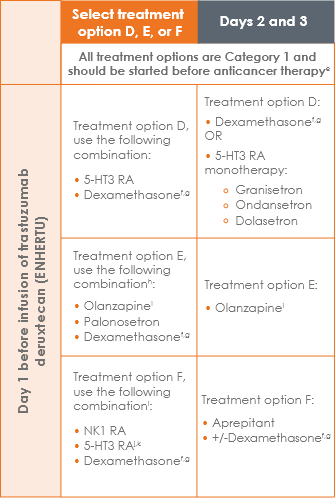
NCCN Guidelines® for Antiemesis lists trastuzumab deruxtecan (ENHERTU) as moderate emetic risk and recommends several prophylactic antiemetic regimens to help decrease potential vomiting5
NCCN prophylactic and management strategies for acute and delayed emesis prevention: Moderate emetic risk parenteral anticancer agents5,b-d
Adapted with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Antiemesis V2.2022. © 2022 National Comprehensive Cancer Network, Inc. All rights reserved.
bFor details regarding recommendations and specific dosing information, please refer to the NCCN Guidelines® for Antiemesis.
cAntiemetic regimens should be chosen based on the drug with the highest emetic risk as well as patient-specific risk factors.
dLorazepam 0.5–1 mg PO or IV or sublingual (SL) every 6 hours as needed on days 1–4. Start with 0.5 mg for patients who are naïve to lorazepam. May be administered with or without H2 blocker or proton pump inhibitor (PPI) if patient exhibits reflux symptoms.
eCategory 1 recommendations indicate uniform NCCN consensus that the intervention is appropriate based on high-level evidence.
fEmerging data and clinical practice suggest dexamethasone doses may be individualized. Higher doses may be considered, especially when an NK1 RA is not given concomitantly. Lower doses, given for shorter durations, or even elimination of dexamethasone on subsequent days (for delayed nausea and emesis prevention) may be acceptable for non-cisplatin regimens based on patient characteristics. If dexamethasone eliminated on subsequent days for delayed nausea and emesis prevention, consider other alternative antiemetics (eg, olanzapine).
gUse of corticosteroid premedications should be avoided with cellular therapies.
hA 3-drug prophylactic regimen (E or F) is recommended for select patients with additional patient-related risk factors or previous treatment failure with a corticosteroid + 5-HT3 RA alone.
iData suggest that a 5-mg dose of olanzapine is efficacious. Consider this dose especially for elderly or over sedated patients.
jIf netupitant/palonosetron or fosnetupitant/palonosetron fixed combination product used, no further 5-HT3 RA is required.
jWhen used in combination with an NK1 RA, there is no preferred 5-HT3 RA.
kNo further 5-HT3 therapy required if palonosetron or granisetron extended-release injection administered, or if granisetron transdermal patch applied, on day 1.
Drug interaction studies2
Clinical studies: There were no clinically meaningful drug-drug interactions between ENHERTU and itraconazole (CYP3A inhibitor), as well as ritonavir (OATP inhibitor)
In vitro studies: DXd did not inhibit common CYP enzymes (CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP3A) nor induce CYP1A2, CYP2B6, or CYP3A
In vitro studies: At clinically relevant concentrations, DXd has a low potential to inhibit the transporters OAT1, OAT3, OCT1, OCT2, OATP1B1, OATP1B3, MATE1, MATE2-K, P-gp, BCRP, or BSEP
WARNING: INTERSTITIAL LUNG DISEASE
Interstitial lung disease (ILD) and pneumonitis, including fatal cases, have been reported with ENHERTU. Monitor for and promptly investigate signs and symptoms including cough, dyspnoea, fever, and other new or worsening respiratory symptoms. Permanently discontinue ENHERTU in all patients with Grade 2 or higher ILD/pneumonitis. Advise patients of the risk and to immediately report symptoms.2
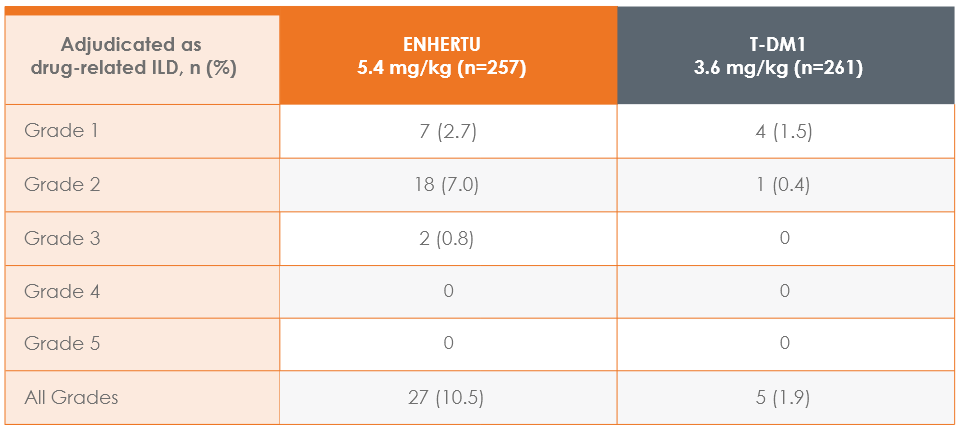
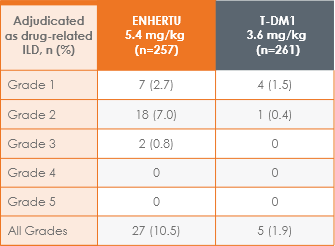
Interstitial Lung Disease (ILD) and Pneumonitis
ILD and pneumonitis, including Grade 5 cases, have been reported with
ENHERTU 5.4 mg/kg (pooled clinical studies; N=573)1,2,a,b
The majority were Grade 1 or 2
In pooled clinical studies in HER2+ mBC, ILD/pneumonitis occurred in 12% of patients; fatal events occurred in 1.4% of patients.
Median time to first onset was 5.5 months (range: 1.1-20.8) with ENHERTU
Primary endpoint demonstrated superiority in progression-free survival as assessed by BICR
lILD includes events that were adjudicated as ILD for ENHERTU: pneumonitis, interstitial lung disease, organizing pneumonia, pneumonia, and pulmonary mass.1
mGrade 5=fatal cases.
Symptom identification and investigation at the first sign of ILD/pneumonitis are key to diagnosis and monitoring
Signs and symptoms of ILD/pneumonitis1
Cough
Dyspnea
Fever
New or worsening respiratory symptoms
Promptly investigate evidence of ILD/pneumonitis6
Diagnosis of ILD/pneumonitis requires exclusion of other causes
Evaluation may include:
High-resolution CT
Pulmonologist consultation
Blood culture and CBC
All events of ILD/pneumonitis, regardless of severity or seriousness, should be followed until resolution including after drug discontinuation
Advise patients of the potential benefits and risks of treatment and to contact HCP immediately to report any of these symptoms
Investigation may be prompted by incidental findings on routine scans when checking for progression or symptomatic findings
For asymptomatic ILD (Grade 1)1
Consider corticosteroid treatment (eg, ≥0.5 mg/ kg/day prednisolone or equivalent)
Interrupt ENHERTU until resolved to Grade 0, then:
If resolved in 28 days or less from date of onset, maintain dose
If resolved in greater than 28 days from date of onset, reduce dose one level .(see Recommended dose reductions for ENHERTU for adverse reactions)
For symptomatic ILD (Grade 2 or greater)1
Promptly initiate systemic corticosteroid treatment (eg, ≥1 mg/kg/day prednisolone or equivalent)
Continue for at least 14 days followed by gradual taper for at least 4 weeks
Permanently discontinue ENHERTU in patients who are diagnosed with any symptomatic ILD/pneumonitis
Talk to your patients to raise awareness and help identify symptoms1
Advise patients to contact their healthcare provider immediately for any of the symptoms shown above
Inform patients of the risks of severe, life-threatening, or fatal ILD
Advise patients to read the approved patient labelling: Download the Medication Guide
Potential questions to ask your patients to help with identification of ILD7,8
✔ Have you been coughing recently? Is it a dry cough?
✔ Have you had any shortness of breath, especially during or after physical activity?
✔ Have you experienced any new breathing or respiratory problems?
✔ If you already have respiratory problems, have they gotten worse?
✔ Have you had a fever?
✔ Have you been feeling tired?
✔ Have you lost weight?
5-HT3, 5-hydroxytryptamine 3; AE, adverse event; ALT, alanine aminotransferase; AR, adverse reaction; AST, aspartate aminotransferase; BCRP, breast cancer resistance protein; BSEP, bile salt export pump; CBC, complete blood count; CT, computed tomography; CYP, cytochrome P450; H2, histamine type 2; HCP, healthcare professional; HER2, human epidermal growth factor receptor 2; ILD, interstitial lung disease; IV, intravenous; mBC, metastatic breast cancer; MATE, multidrug and toxic compound extrusion; MedDRA, Medical Dictionary for Regulatory Activities; NCCN, National Comprehensive Cancer Network; NCI-CTCAE, National Cancer Institute-Common Terminology Criteria for Adverse Events; NK1, neurokinin-1; OAT, organic anion transporter; OATP, organic anion transporting polypeptide; OCT, organic cation transporter; P-gp, P-glycoprotein; PO, by mouth; PT, preferred term; RA, receptor antagonist; SAE, serious adverse event; SMQ, Standardised MedDRA Query; T-DM1, ado-trastuzumab emtansine; TEAE, treatment-emergent adverse event.
References:
1. Cortés J, Kim SB, Chung WP, et al. Trastuzumab deruxtecan (T-DXd) vs trastuzumab emtansine (T-DM1) in patients with HER2+ metastatic breast cancer: results of the randomized, phase 3 study DESTINY-Breast03. Presented at: European Society for Medical Oncology; September 16-21, 2021.
2. ENHERTU. Summary of Product Characteristics.
3. Modi S, Yamashita T, Park YH, et al. Trastuzumab deruxtecan in previously treated HER2-positive breast cancer. N Engl J Med. 382(7):610-621.
4. Supplement to: Cortés J, Kim S-B, Chung W-P, et al. Trastuzumab deruxtecan versus trastuzumab emtansine for breast cancer. N Engl J Med 2022;386:1143-54.
5. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Antiemesis V2.2022. © National Comprehensive Cancer Network, Inc. 2022. All rights reserved. Accessed September, 2022. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
6. Modi S, Saura C, Yamashita T, et al. Trastuzumab deruxtecan in previously treated HER2-positive breast cancer [supplementary appendix]. N Engl J Med. 2020;382(7):610-621.
7. Ripley BA, Kelil T, Gill RR. Deciphering drug-induced interstitial lung disease: a mechanistic approach. Appl Radiol. 2016;45(4):9-18.
8. Kubo K, Azuma A, Kanazawa M, et al; Japanese Respiratory Society Committee. Consensus statement for the diagnosis and treatment of drug-induced lung injuries. Respir Invest. 2013;51(4):260-277.