DESTINY-Breast03 Trial
Trastuzumab deruxtecan (T-DXd; DS-8201) versus trastuzumab emtansine (T-DM1) in patients with HER2 positive metastatic breast cancer
Updated results of the randomized, open-label, multicenter, phase 3 study.
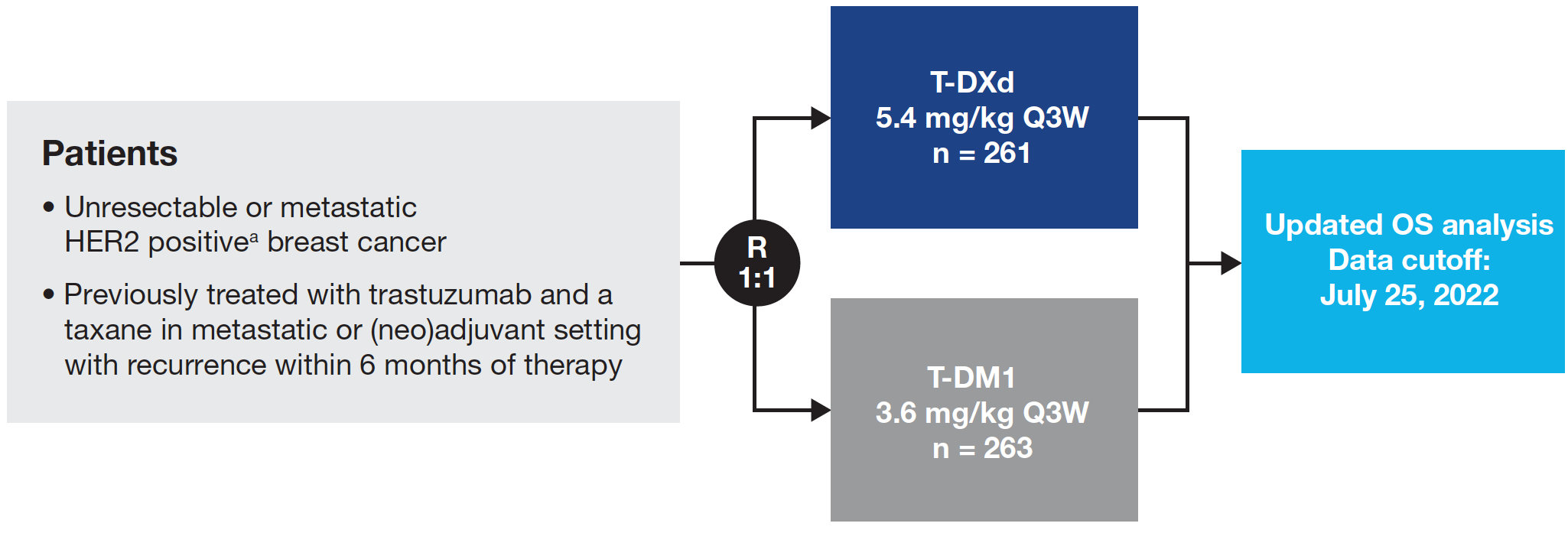
DESTINY-Breast03 Study Design1,2
Endpoints1,2
Primary Endpoint
- PFS (BICR)
Key Secondary Endpoint
- OSb
Other Endpoints
- ORR (BICR and investigator)
- DoR (BICR)
- PFS (investigator)
- CBR (BICR)
- PFS2 (investigator)
- Safety
Analysis Population1,2
Adult patients with HER2 positive unresectable and/or metastatic BC who were previously treated with trastuzumab and taxane in the advanced/metastatic setting or progressed during or within 6 months after neoadjuvant/adjuvant treatment involving trastuzumab and a taxane. Herein, we present updated OS analysis results with a data cutoff of July 25, 2022.
Enrollment1,2
A total of 524 patients with HER2 positive metastatic BC were randomly assigned 1:1 to receive T-DXd 5.4 mg/kg (n = 261) or T-DM1 3.6 mg/kg (n = 263). Together these patients comprised the full analysis set.
Please check local regulations for approval status for trastuzumab deruxtecan. Information regarding this investigational product should, under no circumstances, be regarded as a recommendation for its use or of its safety or efficacy.
aHER2 IHC 3+ or IHC 2+/ISH+ based on central confirmation. b80% powered at 2-sided significance level of 5%.
BC, breast cancer; BICR, blinded independent central review; CBR, clinical benefit rate; DoR, duration of response; HER2, human epidermal growth factor receptor 2; IHC, immunohistochemistry; ISH, in situ hybridization; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; PFS2, progression-free survival on the next line of therapy; Q3W, every 3 weeks; R, randomized; T-DM1, trastuzumab emtansine; T-DXd, trastuzumab deruxtecan.
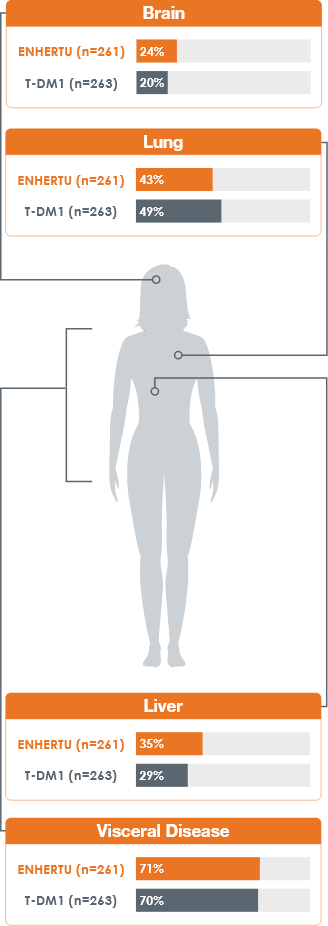
Key Findings
Patient Characteristics1,2
- Patient characteristics were balanced across the 2 arms
- Patients in both arms had a median of 2 prior lines of therapy in the metastatic setting
- 62.1% and 60.1% of patients previously received pertuzumab in the T-DXd and T-DM1 arms, respectively
Efficacy1,2
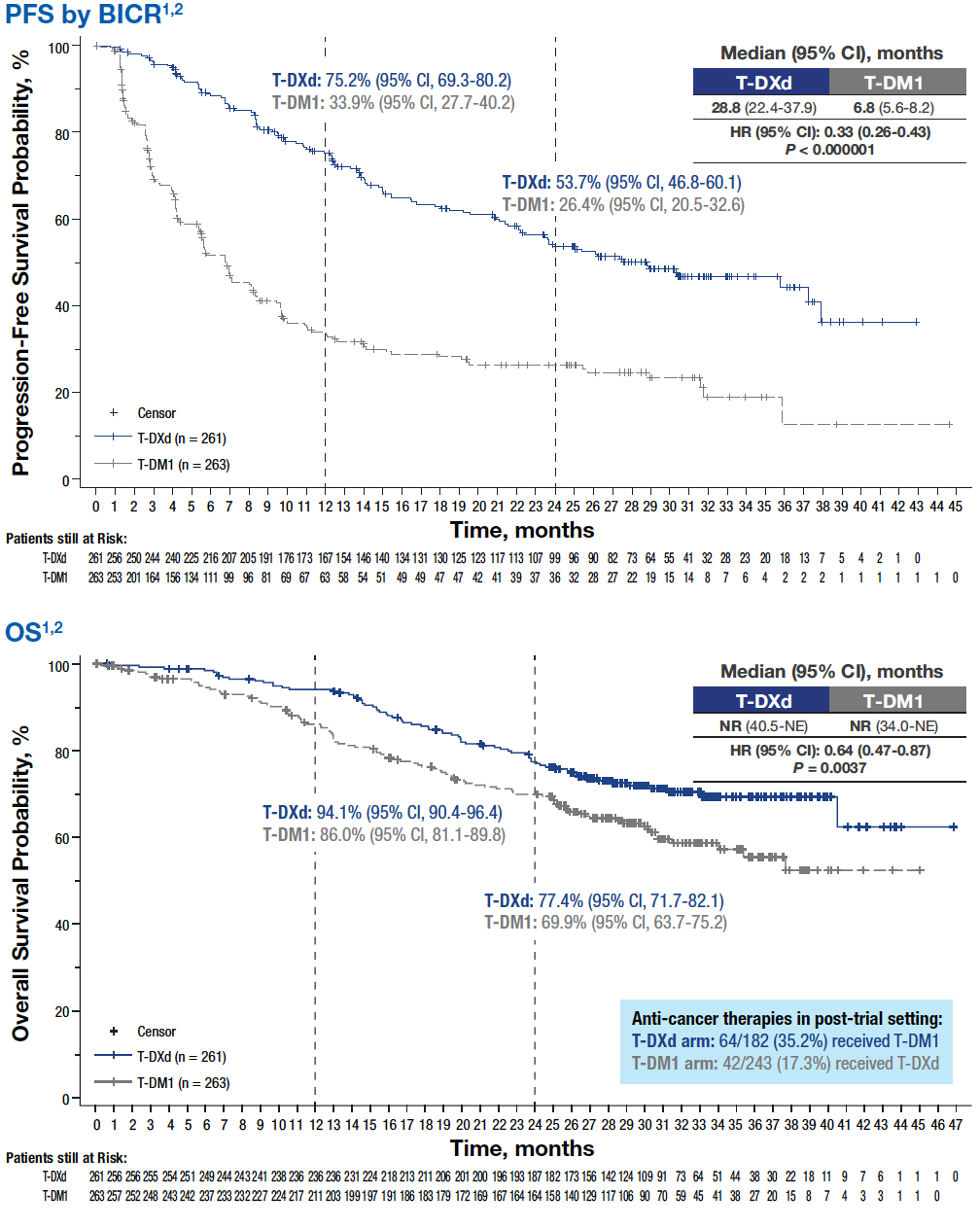
- Treatment with T-DXd resulted in a clinically meaningful and statistically significant improvement in PFS by BICR
- Median PFS assessed by BICR was 28.8 months with T-DXd (95% CI, 22.4-37.9) versus 6.8 months (95% CI, 5.6-8.2) with T-DM1; HR, 0.33 (95% CI, 0.26-0.43); nominal P < 0.000001
- Landmark PFS rates were greater for T-DXd versus T-DM1 at 12 months (75.2% vs 33.9%) and 24 months (53.7% vs 26.4%)
- T-DXd significantly reduced the risk of death by 36% versus T-DM1 (HR, 0.64)
- Median OS was NR (95% CI, 40.5-NE) for T-DXd versus NR (95% CI, 34.0-NE) for T-DM1; HR, 0.64 (95% CI, 0.47-0.87; P = 0.0037)
- Landmark OS rates were greater for T-DXd versus T-DM1 at 12 months (94.1% vs 86.0%) and 24 months (77.4% vs 69.9%)
- OS benefit was consistent across key subgroups
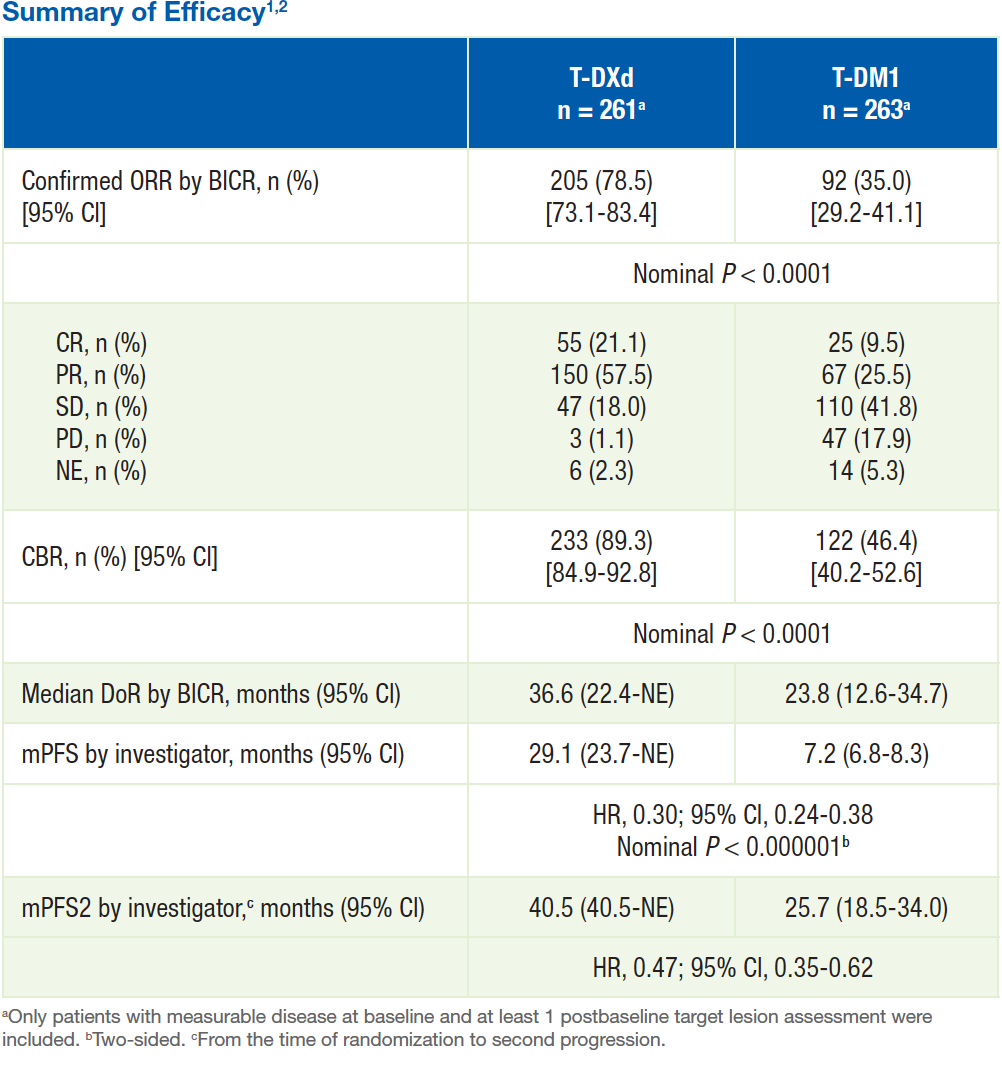
- ORR by BICR was greater for T-DXd versus T-DM1 (78.5% vs 35.0%)
- 55 (21.1%) T-DXd-treated patients versus 25 (9.5%) T-DM1-treated patients experienced CR, and most patients treated with T-DXd experienced PR (150 [57.5%]) versus 67 (25.5%) with T-DM1
- 3 (1.1%) T-DXd-treated patients versus 47 (17.9%) T-DM1-treated patients experienced PD
- CBR was greater for T-DXd versus T-DM1 (89.3% vs 46.4%)
Safety1,2
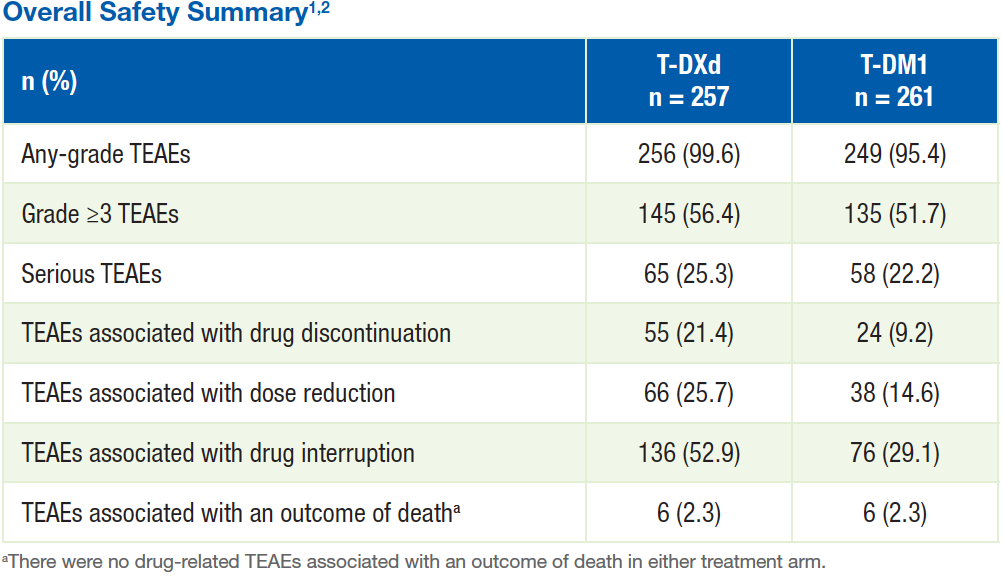
- With a longer treatment duration, T-DXd continues to demonstrate a manageable and tolerable safety profile
- Median treatment duration with T-DXd was 18.2 months (range, 0.7-44.0) versus 6.9 months (range, 0.7-39.3) with T-DM1
- 19.8% and 25.3% of patients treated with T-DXd experienced a drug-related TEAE associated with drug discontinuation or dose reduction, respectively
- 6.5% and 14.6% of patients treated with T-DM1 experienced a drug-related TEAE associated with drug discontinuation or dose reduction, respectively
- The most common TEAEs in ≥20% of patients of any grade were mostly gastrointestinal and hematological in nature with T-DXd, and included nausea (T-DXd, 77.0%; T-DM1, 30.3%), vomiting (T-DXd, 51.8%; T-DM1, 10.7%), and alopecia (T-DXd, 39.7%; T-DM1, 3.4%)
- Adjudicated ILD/pneumonitis related to T-DXd was observed in 39 patients (15.2%) and most cases were grade 1 or 2 (grade 1, n = 11; grade 2, n = 26; grade 3, n = 2; no grade 4/5 events)
- Adjudicated ILD/pneumonitis related to T-DM1 was observed in 8 patients (3.1%) and most cases were grade 1 or 2 (grade 1, n = 4; grade 2, n = 3; grade 3, n = 1, no grade 4/5 events)
Overall Safety Summary1,2
Please check local regulations for approval status for trastuzumab deruxtecan. Information regarding this investigational product should, under no circumstances, be regarded as a recommendation for its use or of its safety or efficacy.
References:
1. Hurvitz S et al. Trastuzumab deruxtecan versus trastuzumab emtansine in patients with HER2-positive metastatic breast cancer: Updated results from DESTINY-Breast03, a randomised, open-label, phase 3 trial. The Lancet. 2022 [in press]. 2. Hurvitz S et al. Trastuzumab deruxtecan versus trastuzumab emtansine in patients with HER2-positive metastatic breast cancer: Updated results of the randomized, phase 3 study DESTINY-Breast03. Presented at the San Antonio Breast Cancer Symposium (SABCS) 2022 Annual Meeting; December 6-10, 2022. Presentation GS2-02.
BICR, blinded independent central review; CBR, clinical benefit rate; CI, confidence interval; CR, complete response; DoR, duration of response; HR, hazard ratio; ILD, interstitial lung disease; mPFS, median progression-free survival; mPFS2, median progression-free survival on the next line of therapy; NE, not evaluable; NR, not reached; ORR, objective response rate; OS, overall survival; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease; T-DM1, trastuzumab emtansine; T-DXd, trastuzumab deruxtecan; TEAE, treatment-emergent adverse event.