Configure Section Container Component
Safety
Pulmicort® has the most extensive nebulised ICS paediatric clinical data on efficacy and safety across all asthma severities and ages
Pulmicort® Respules® is the only inhaled corticosteroid available for the treatment of asthmatic children from the age of 6 months.4
- Long-term use of nebulized budesonide has no apparent influence on the HPA* function in children with asthma, according to GINA recommended scheme.1
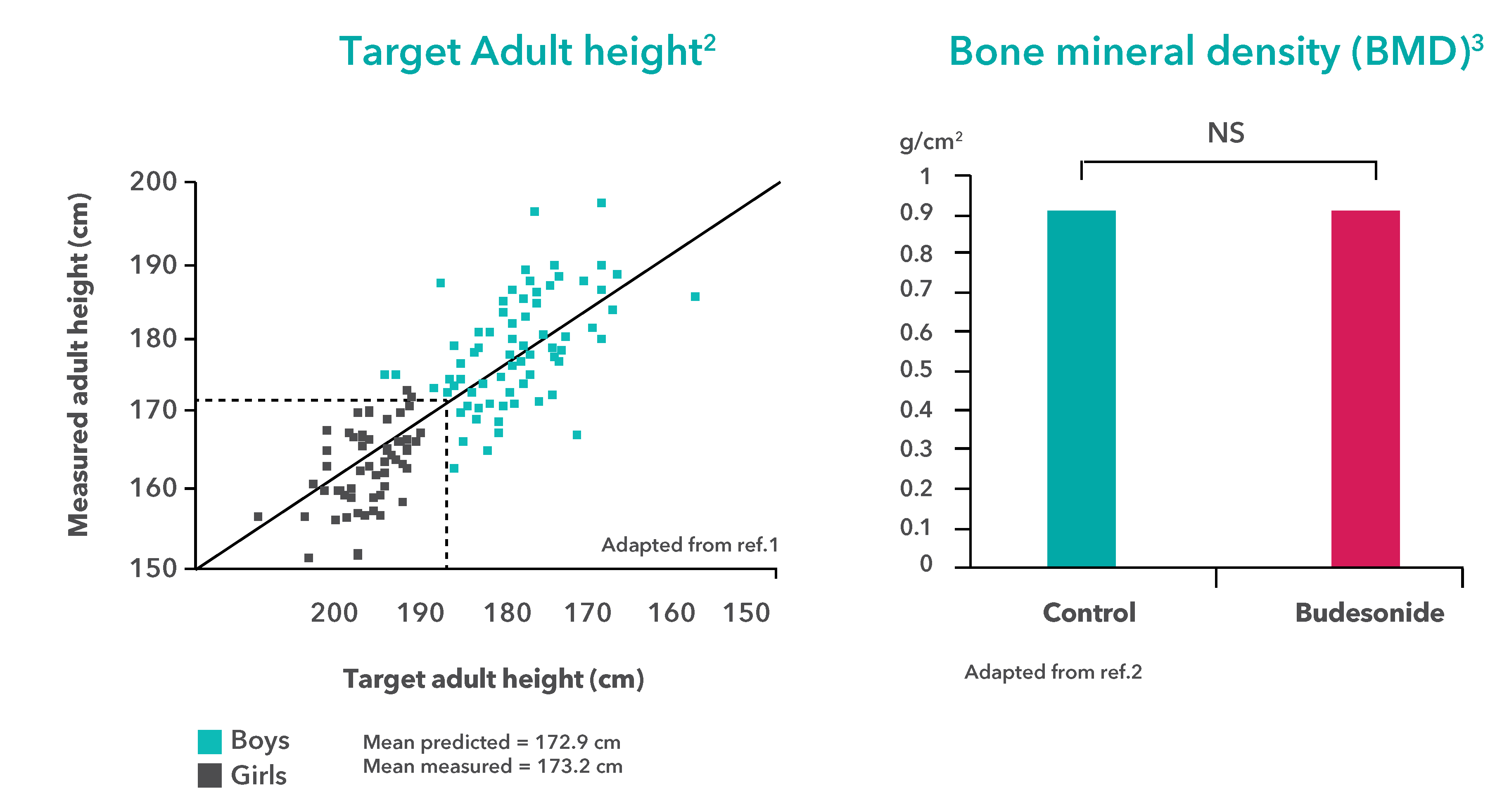
- Long-term treatment with inhaled budesonide* DID NOT affect growth or development in children aged 3 to 13 years old.†,4
Configure Image Component
References
- Zhan JY et al. Influenece of Nebulized Corticosteroids on Hyopthalamic-Pituitary-Adrenal Axia in children with Asthma J Appl Clin 6-24:1244;2009.
- Agertoft, Lone and SØren Pedersen. “Effect Of Long-Term Treatment With inhaled Budesonide On Adult Height In Children With Asthma”. New England Journal of Medicine 343.15 (2000): 1064-1069. Web.
- AGERTOFT, LONE and SØREN PEDERSEN. “Bone Mineral Desnsity in Children With Asthma Receiving Long-Term Treatment With inhaled Budesonide”. American Journal of Respiratory and Critical Care Medicine 157.1 (1998): 178-183. Web.
- Pulmicort® Respules® SmPC.
SA-3644
Modal Window Component Section Begins
Modal Window Component Section Ends
Configure Footer Reference Component