Mechanism of Action
See how ENHERTU delivers the cytotoxic agent to HER2+ cells1,2
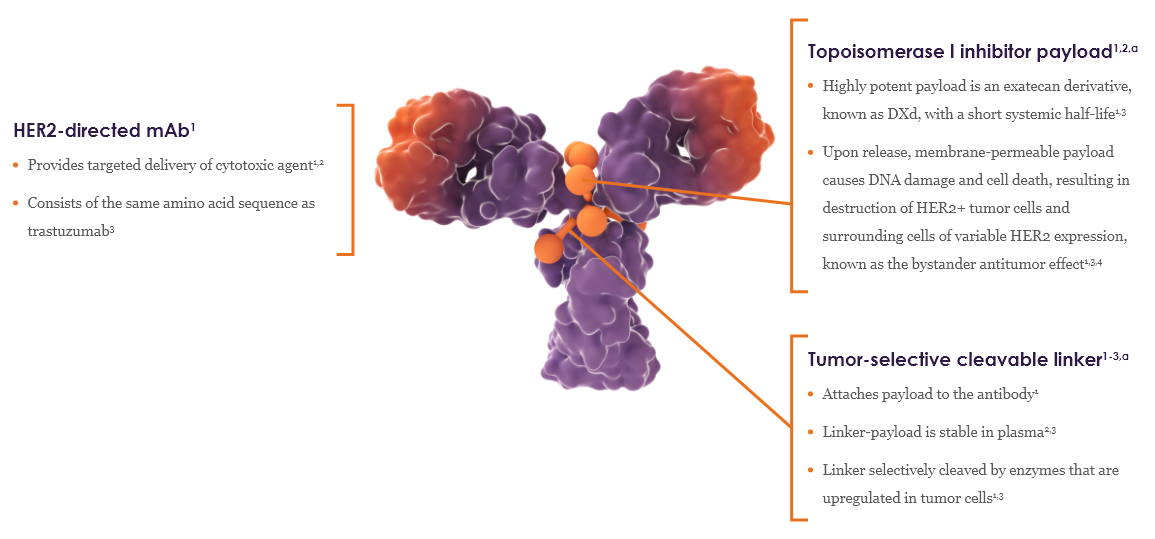
ENHERTU is a specifically engineered HER2-directed antibody-drug conjugate (ADC)1,2
HER2-directed mAb1
- Provides targeted delivery of cytotoxic agent1,2
- Consists of the same amino acid sequence as trastuzumab3
Topoisomerase I inhibitor payload1,2,a
- Highly potent payload is an exatecan derivative, known as DXd, with a short systemic half-life1,3
- Upon release, membrane-permeable payload causes DNA damage and cell death, resulting in destruction of HER2+ tumor cells and surrounding cells of variable HER2 expression, known as the bystander antitumor effect1,3,4
Tumor-selective cleavable linker1-3,a
- Attaches payload to the antibody1
- Linker-payload is stable in plasma2,3
- Linker selectively cleaved by enzymes that are upregulated in tumor cells1,3
ENHERTU has a homogeneous and high drug-to-antibody ratio of ~8 molecules of cytotoxic agent per antibody1-3,a,b
aBased on in vitro and in vivo non-clinical studies. The clinical relevance of these features is under investigation.
bADCs are a mixture of molecules in which the DAR is variable. Homogeneity of DAR refers to a mixture where there is low variability of DAR; the payload number per antibody falls into a narrow range.3
Dosage, Preparation, and Administration
What is the dosage for ENHERTU and how is it given?
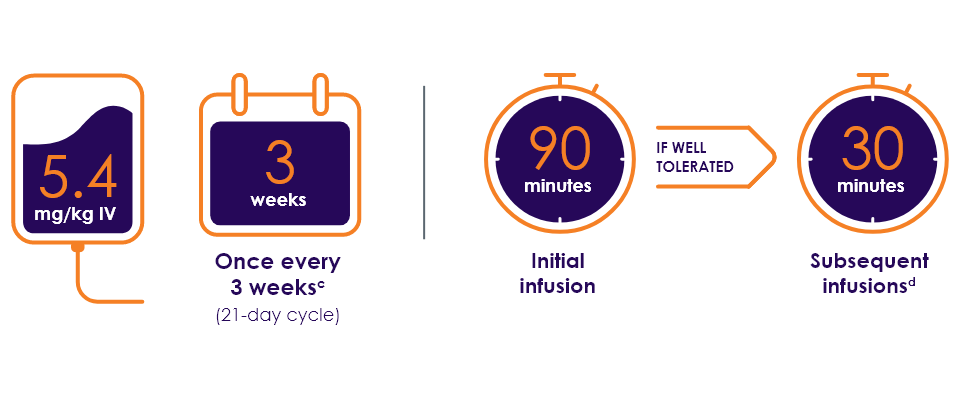
Recommended dosage and schedule1
- Permanently discontinue ENHERTU in case of severe infusion reactions
- Slow or interrupt the infusion rate if the patient develops infusion-related symptoms
- Do not substitute ENHERTU with trastuzumab or trastuzumab emtansine
ENHERTU should be prescribed by a physician and administered under the supervision of a healthcare professional experienced in the use of anticancer medicinal products.
Dosage forms and strengths1
For infusion: 100 mg of ENHERTU as a white to yellowish-white lyophilised powder in a single-dose vial for reconstitution and further dilution.
cUntil disease progression or unacceptable toxicity.1
dAntiemetics may be administered in accordance with local medical practice as per patient tolerance for prophylaxis or management.1
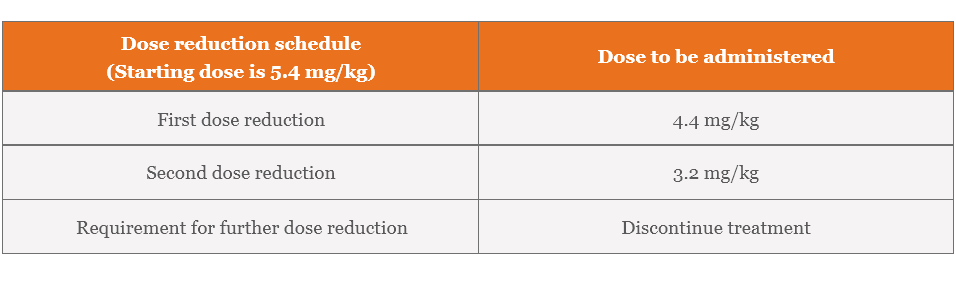
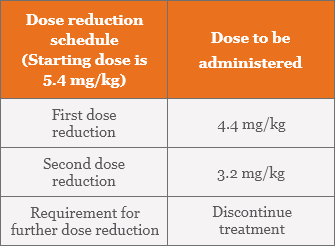
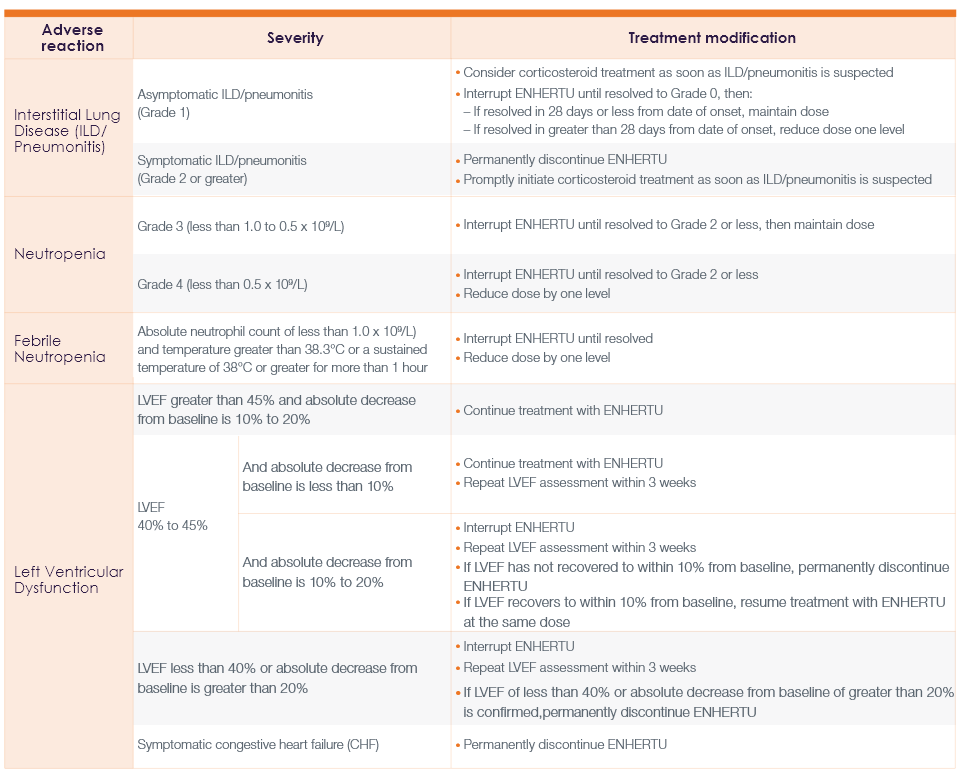
Recommended dose reductions for ENHERTU for adverse reactions1
Management of adverse reactions may require temporary interruption, dose reduction, or treatment discontinuation of ENHERTU per guidelines provided in the tables below.
Dose modifications for adverse reactions1
How do I prepare ENHERTU for use?
- In order to prevent medication errors, check the vial labels to ensure that the drug being prepared and administered is ENHERTU and not trastuzumab or trastuzumab emtansine1
- ENHERTU requires reconstituting and diluting prior to intravenous infusion4
- Use appropriate aseptic technique1
- ENHERTU is a cytotoxic drug. Follow applicable special handling and disposal procedures1,5
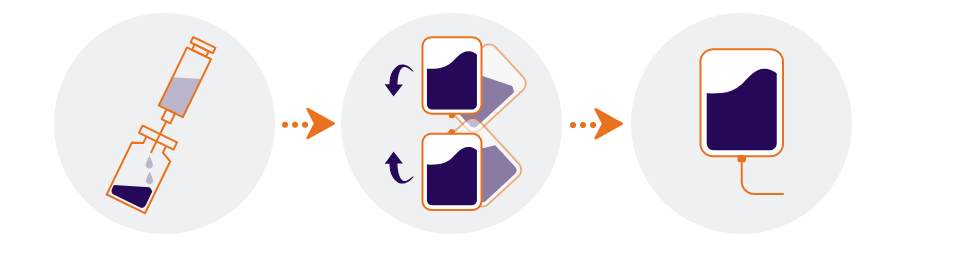
Steps to prepare ENHERTU for use

How do I reconstitute ENHERTU?1
- Reconstitute immediately before dilution
- More than one vial may be needed for a full dose. Calculate the dose (mg), the total volume of reconstituted ENHERTU solution required, and the number of vial(s) of ENHERTU needed
Reconstitute each 100 mg vial by using a sterile syringe to slowly inject 5 mL of water for injection into each vial to obtain a final concentration of 20 mg/mL - Swirl the vial gently until completely dissolved. Do not shake
- Inspect the reconstituted solution for particulates and discolouration prior to administration. The solution should be clear and colourless to light yellow. Do not use if visible particles are observed or if the solution is cloudy or discoloured
- If not used immediately, store the reconstituted ENHERTU vials in a refrigerator at 2–8°C for up to 24 hours from the time of reconstitution, protected from light. Do not freeze
- The reconstituted product contains no preservative and is intended for single use only

How do I dilute ENHERTU?1
- Dilute the calculated volume of reconstituted ENHERTU in an infusion bag containing 100 mL of 5% glucose solution. Do not use sodium chloride solution. An infusion bag made of polyvinylchloride or polyolefin (copolymer of ethylene and polypropylene) is recommended
- Gently invert the infusion bag to thoroughly mix the solution. Do not shake
- Cover the infusion bag to protect it from light
- If not used immediately, store at room temperature for up to 4 hours, including preparation and infusion, or in a refrigerator at 2–8°C for up to 24 hours, protected from light. Do not freeze
- Discard any unused portion left in the vial

How do I give ENHERTU?1
- If the prepared infusion solution was stored refrigerated (2–8°C), it is recommended that the solution be allowed to equilibrate to room temperature prior to administration, protected from light
- Administer ENHERTU as an intravenous infusion only with a 0.20 or 0.22 micron in-line polyethersulfone (PES) or polysulfone (PS) filter
- The initial dose should be administered as a 90-minute intravenous infusion. If the prior infusion was well tolerated, subsequent doses of ENHERTU may be administered as 30-minute infusions. Do not administer as an intravenous push or bolus
- Do not mix ENHERTU with other medicinal products or administer other medicinal products through the same intravenous line
DAR, drug-to-antibody ratio; DNA, deoxyribonucleic acid; HER2, human epidermal growth factor receptor 2; IV, intravenous; LVEF, left ventricular ejection fraction; mAb, monoclonal antibody; mBC, metastatic breast cancer; NCI-CTCAE, National Cancer Institute–Common Terminology Criteria for Adverse Events.
References:
1. ENHERTU. Summary of Product Characteristics.
2. Ogitani Y, Aida T, Hagihara K, et al. DS-8201a, a novel HER2-targeting ADC with a novel DNA topoisomerase I inhibitor, demonstrates a promising antitumor efficacy with differentiation from T-DM1. Clin Cancer Res. 2016;22(20):5097-5108.
3. Nakada T, Sugihara K, Jikoh T, Abe Y, Agatsuma T. The latest research and development into the antibody–drug conjugate, [fam-] trastuzumab deruxtecan (DS-8201a), for HER2 cancer therapy. Chem Pharm Bull (Tokyo). 2019;67(3):173-185.
4. Ogitani Y, Hagihara K, Oitate M, Naito H, Agatsuma T. Bystander killing effect of DS-8201a, a novel anti-human epidermal growth factor receptor 2 antibody-drug conjugate, in tumours with human epidermal growth factor receptor 2 heterogeneity. Cancer Sci. 2016;107(7):1039-1046.
5. OSHA Hazardous Drugs. OSHA. Available at http://www.osha.gov/SLTC/hazardousdrugs/index.html. Accessed September, 2022.