TTR SUPPRESSION
WAINUA provides rapid and sustained TTR
suppression between doses in adults with hATTR-PN*1,2
LSM % CHANGE FROM BASELINE IN SERUM TTR1,2
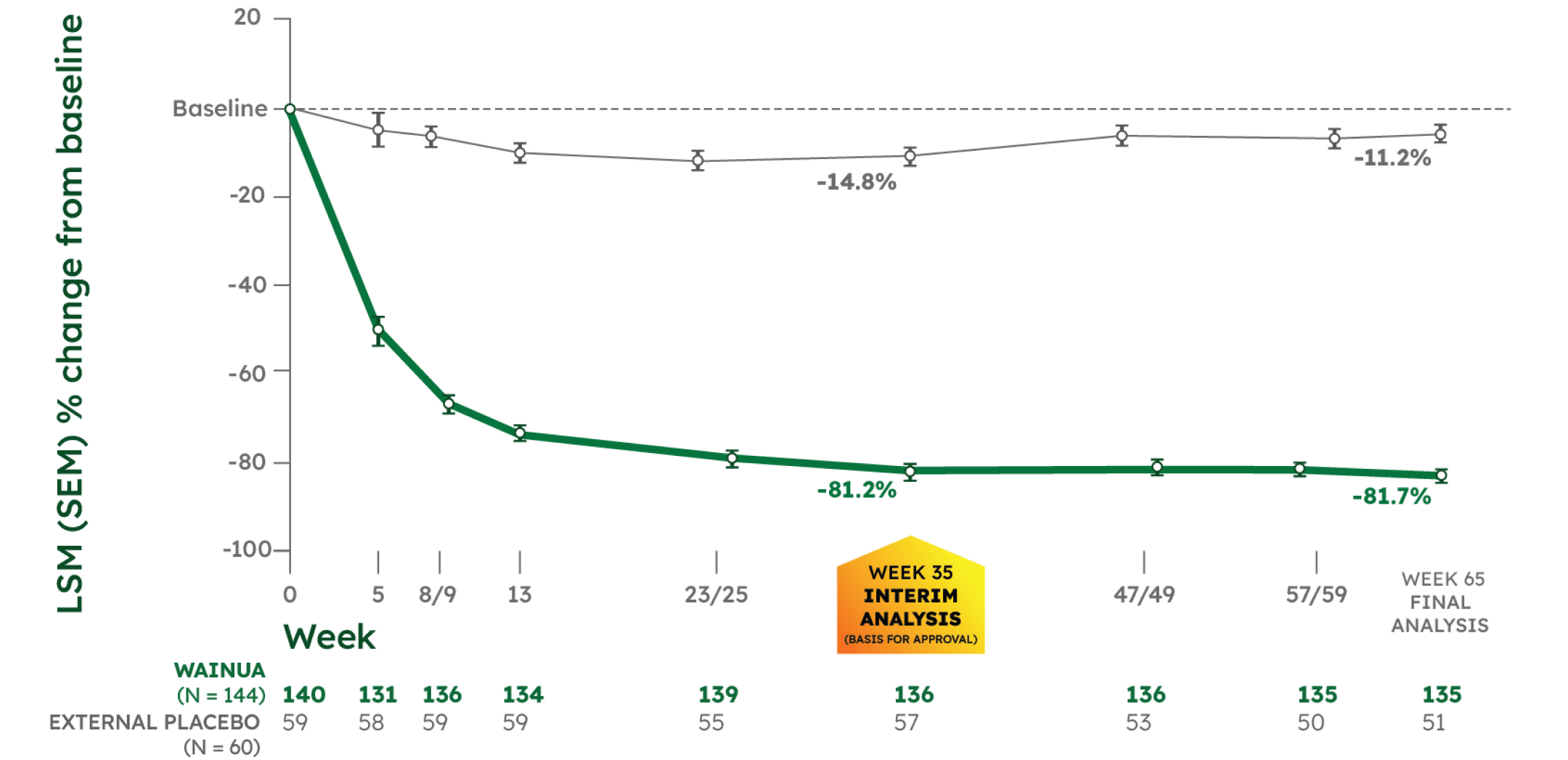
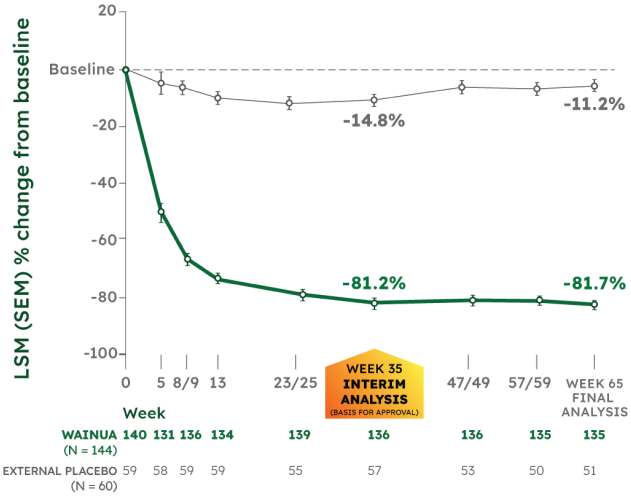
CO-PRIMARY ENDPOINT:
- -66.4% significant difference with WAINUA vs placebo at Week 35 (95% CI, -71.4 to -61.5), P<0.001
- -70.4% difference with WAINUA vs placebo at Week 65 (95% CI, -75.2 to -65.7)
NEURO-TTRansform was a phase 3, randomized, open-label trial. Data presented from baseline to Week 35 are from the interim analysis; data beyond Week 35 are from the final analysis. Because the primary and key secondary endpoints were statistically significant at Week 35, no further formal statistical analyses were performed at Week 65/66. See study design.1-4
Reductions in serum TTR levels were sustained through Week 85 in adults with hATTR-PN1,2
Post hoc analysis
MEAN % CHANGE FROM BASELINE IN SERUM TTR1,2
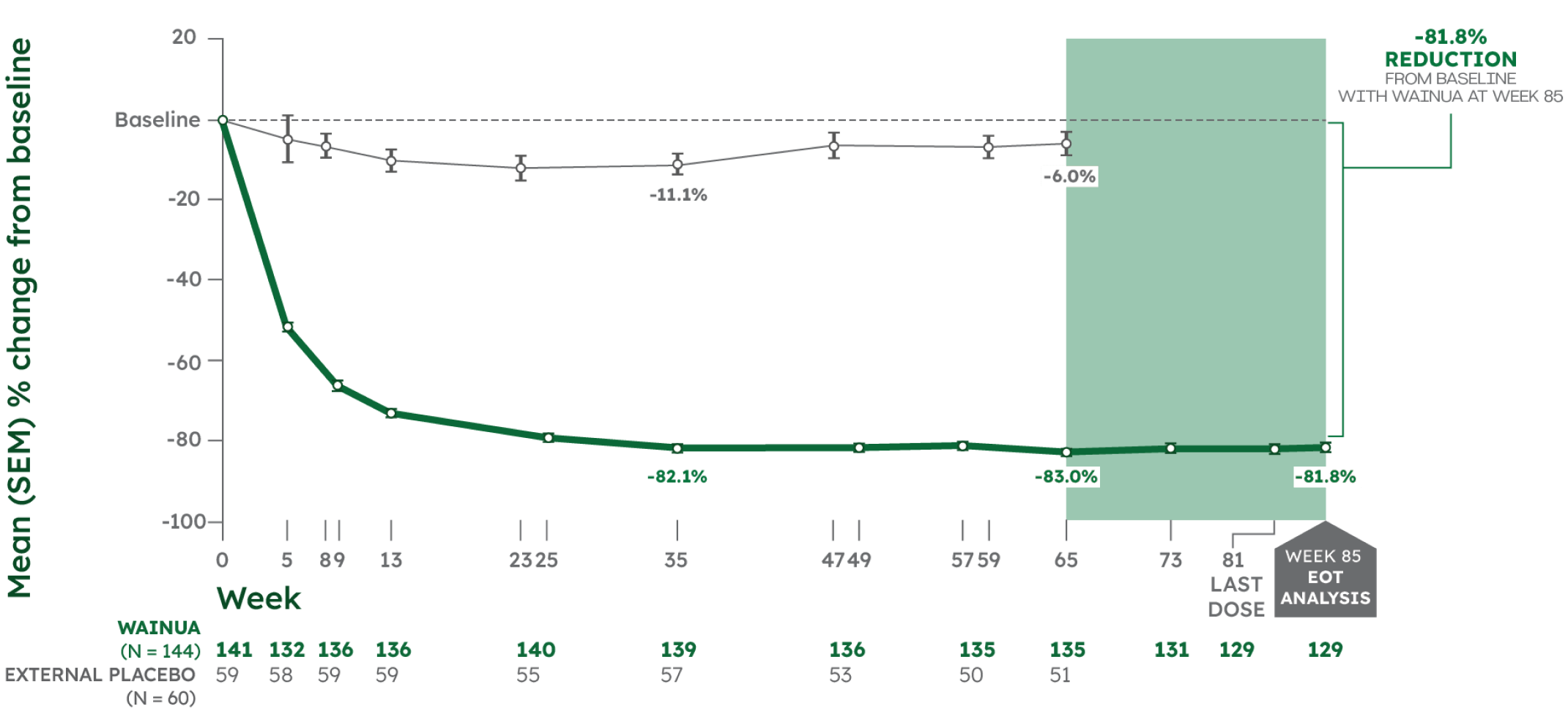
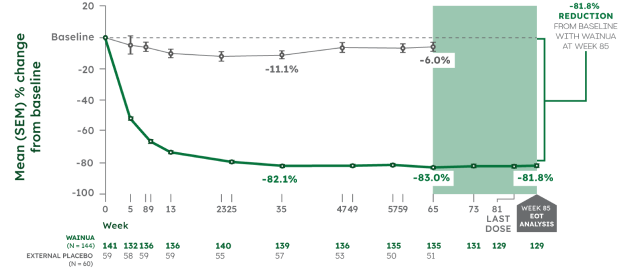
Note: From Week 66 to Week 85, there was no external placebo group because NEURO-TTR concluded at Week 66. Change in serum TTR levels was assessed as a post hoc outcome at Week 85. No formal statistical analyses were performed at Week 85.2
*Serum TTR level was first measured at Week 5. WAINUA 45 mg SC was dosed at Week 1 and once every 4 weeks thereafter. Serum TTR levels were drawn at trough drug levels predose, except at Week 35 and Week 85, which were drawn between doses.2,3
Discover the powerful efficacy of WAINUA
Explore the NEURO-TTRansform study design
FOOTNOTES
EOT, end of treatment; hATTR-PN, polyneuropathy of hereditary transthyretin-mediated amyloidosis; LSM, least-squares mean; SC, subcutaneous; SEM, standard error of mean; TTR, transthyretin.
REFERENCES
- 1. Data on File, REF-205487, AZPLP.
- 2. Coelho T, Marques W Jr, Dasgupta NR, et al. Eplontersen for hereditary transthyretin amyloidosis with polyneuropathy [article and supplementary online content]. JAMA. 2023;330(15):1448-1458.
- 3. WAINUA® (eplontersen) [prescribing information]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2025.
- 4. Coelho T, Ando Y, Benson MD, et al. Design and rationale of the global phase 3 NEURO-TTRansform study of antisense oligonucleotide AKCEA-TTR-LRx (ION-682884-CS3) in hereditary transthyretin-mediated amyloid polyneuropathy. Neurol Ther. 2021;10(1):375-389.
IMPORTANT SAFETY INFORMATION
WARNINGS AND PRECAUTIONS
- Reduced Serum Vitamin A Levels and Recommended Supplementation WAINUA leads to a decrease in serum vitamin A levels. Supplement with recommended daily allowance of vitamin A. Refer patient to an ophthalmologist if ocular symptoms suggestive of vitamin A deficiency occur.
ADVERSE REACTIONS
Most common adverse reactions (≥9% in WAINUA-treated patients) were vitamin A decreased (15%) and vomiting (9%).
INDICATION
WAINUA injection, for subcutaneous use, 45 mg is indicated for the treatment of the polyneuropathy of hereditary transthyretin-mediated amyloidosis
in adults.


