ADDITIONAL ENDPOINTS
Key secondary and prespecified exploratory endpoints in the NEURO-TTRansform trial
The power to prevent nutritional decline1
WAINUA demonstrated significant improvement in mBMI, a measure of nutritional status, from baseline vs external placebo at Week 651
Key secondary endpoint: LSM change in mBMI from baseline1
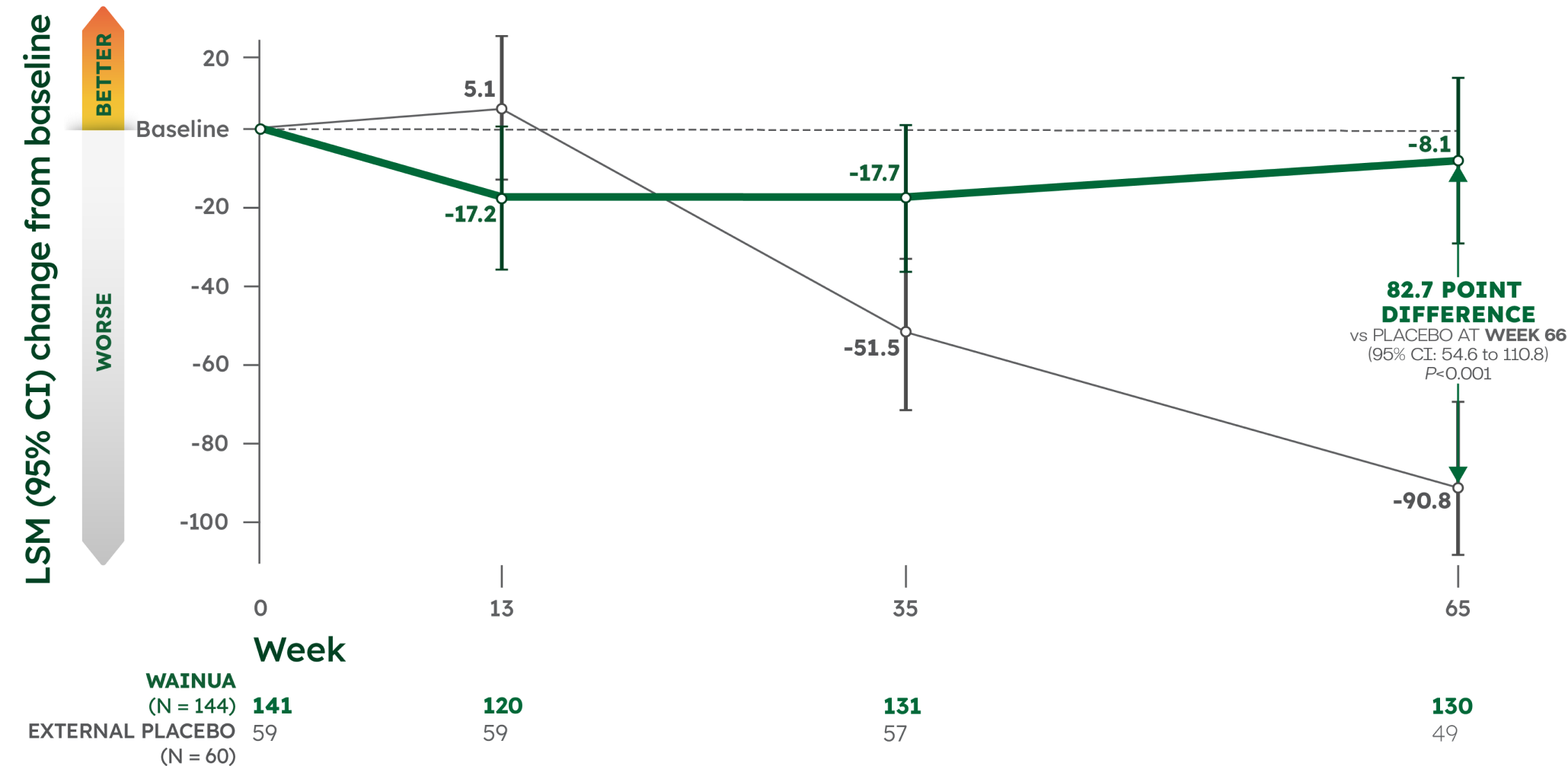
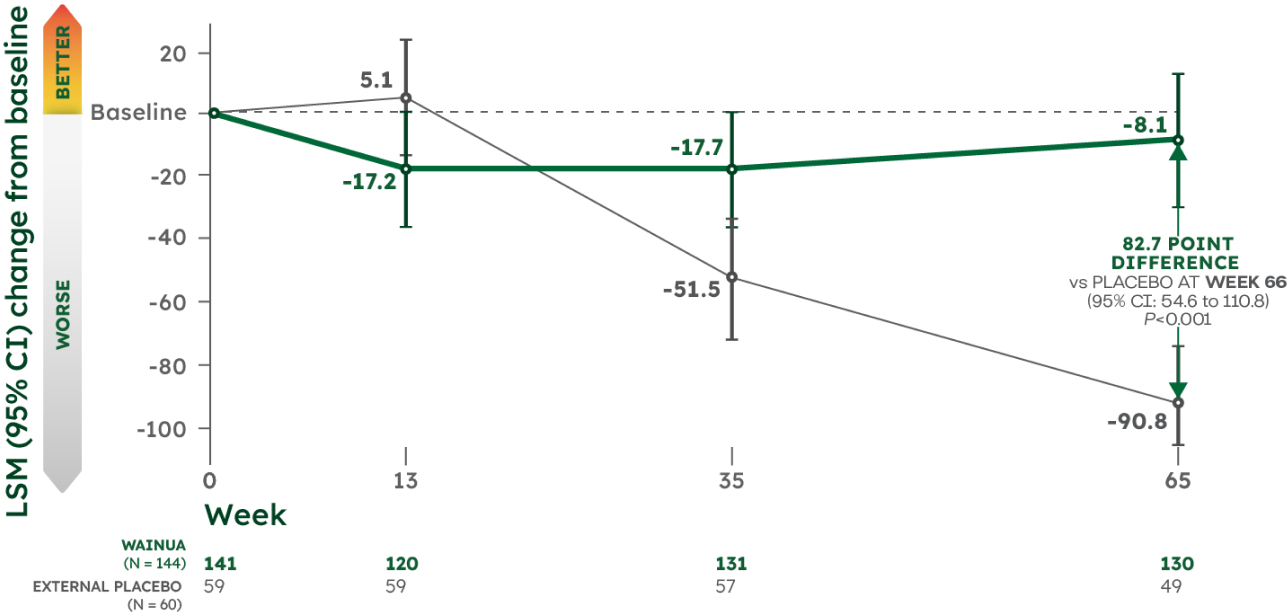
mBMI (modified body mass index=body mass index [kg/m2] × serum albumin [g/L]) was measured as a secondary endpoint at Week 65 and is a validated assessment of nutritional status. See study design.1
UNDERSTANDING mBMI:
mBMI is a validated measure that accounts for volume overload by including the serum albumin levels, and is defined as body mass index (BMI) × albumin, with higher scores indicating better nutritional status.2,3

mBMI = BMI (kg/m2) × serum albumin (g/L)
Improvement in COMPASS-31 score, a measurement of autonomic neuropathy symptoms that include GI and orthostatic intolerance2
Prespecified exploratory endpoint: mean (+/- SE) change from baseline with WAINUA in COMPASS-31 total score at Week 81*†‡1,2


Change from baseline in COMPASS-31 total score (mean +/- SE) at Week 81 was measured as a prespecified exploratory endpoint. This analysis did not include the external placebo group, as COMPASS-31 scores were not collected in the NEURO-TTR trial. See study design.1,2
*COMPASS-31 total score at baseline in patients treated with WAINUA (n=141) was 19.4 (SD, 11.3).2
†COMPASS-31 is a 31-item questionnaire that evaluates autonomic function across 6 domains. Final scores range from 0 to 100, with higher scores indicating worse autonomic dysfunction.1
‡This analysis did not include the external placebo group, as COMPASS-31 scores were not collected in the NEURO-TTR trial.2
The COMPASS-31 questionnaire is recommended for broad assessment of the severity and extent of a range of specific autonomic symptoms4
COMPASS-31 is a 31-item, self-administered questionnaire that evaluates autonomic function across 6 domains5:
Gastrointestinal
Covers symptoms like early satiety, bloating, vomiting, and bowel movement irregularities
Orthostatic intolerance
Evaluates symptoms like dizziness or lightheadedness upon standing
Vasomotor
Assesses symptoms related to blood vessel function like skin color changes
Secretomotor
Assesses problems related to sweating and dryness of eyes or mouth
Pupillomotor
Assesses light sensitivity and focusing difficulties
Bladder
Evaluates urinary symptoms, including incontinence and difficulty in voiding
Explore the safety profile of WAINUA
Learn more about once-monthly self-administration
FOOTNOTES
COMPASS-31, Composite Autonomic Symptom Score-31; GI, gastrointestinal; LSM, least-squares mean; mBMI, modified body mass index; SD, standard deviation; SE, standard error.
REFERENCES
- 1. Coelho T, Marques W Jr, Dasgupta NR, et al. Eplontersen for hereditary transthyretin amyloidosis with polyneuropathy [article and supplementary online content]. JAMA. 2023;330(15):1448-1458.
- 2. Wixner J, Berk J, Adams D, et al. Eplontersen improves autonomic neuropathy symptoms in hereditary ATTR: an analysis from NEURO-TTRansform. Presented at: 4th International ATTR Amyloidosis Meeting; November 2-3, 2023; Madrid, Spain.
- 3. Kittleson MM, Ruberg FL, Ambardekar AV, et al; Writing Committee. 2023 ACC Expert Consensus Decision Pathway on comprehensive multidisciplinary care for the patient with cardiac amyloidosis: a report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol. 2023;81(11):1076-1126.
- 4. Wixner J, Berk JL, Adams D, et al. Effects of eplontersen on symptoms of autonomic neuropathy in hereditary transthyretin-mediated amyloidosis: secondary analysis from the NEURO-TTRansform trial. Amyloid. 2025;32(1):29-38.
- 5. Sletten DM, Suarez GA, Low PA, Mandrekar J, Singer W. COMPASS 31: a refined and abbreviated composite autonomic symptom score [article and supplementary appendix]. Mayo Clin Proc. 2012;87(12):1196-1201.
IMPORTANT SAFETY INFORMATION
WARNINGS AND PRECAUTIONS
- Reduced Serum Vitamin A Levels and Recommended Supplementation WAINUA leads to a decrease in serum vitamin A levels. Supplement with recommended daily allowance of vitamin A. Refer patient to an ophthalmologist if ocular symptoms suggestive of vitamin A deficiency occur.
ADVERSE REACTIONS
Most common adverse reactions (≥9% in WAINUA-treated patients) were vitamin A decreased (15%) and vomiting (9%).
INDICATION
WAINUA injection, for subcutaneous use, 45 mg is indicated for the treatment of the polyneuropathy of hereditary transthyretin-mediated amyloidosis
in adults.


