MECHANISM OF ACTION
WAINUA is the only ASO with LICA technology approved for hATTR-PN, enabling direct binding to hepatocytes1-5

WAINUA mechanism of action

Liver-specific targeting1,2,6
A silencer that directly binds to hepatocytes, reducing off-target exposure through GalNAc conjugation
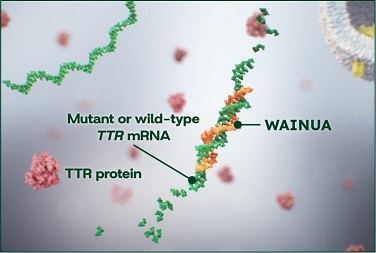
TTR mRNA degradation2,6
Degradation of both mutant and wild-type TTR mRNA
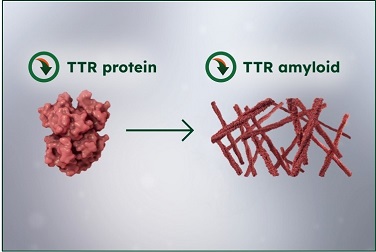
Reduction of TTR protein production2,6
Resulting in reduced serum TTR protein and subsequent amyloid formation
Play this video to understand how WAINUA works
Learn how WAINUA can affect serum TTR levels
Explore the NEURO-TTRansform study design
FOOTNOTES
ASGPR, asialoglycoprotein receptor; GalNAc, N-acetyl galactosamine; hATTR-PN, polyneuropathy of hereditary transthyretin-mediated amyloidosis; LICA, ligand-conjugated antisense; mRNA, messenger ribonucleic acid; TTR, transthyretin.
REFERENCES
- 1. Viney NJ, Guo S, Tai L-J, et al. Ligand conjugated antisense oligonucleotide for the treatment of transthyretin amyloidosis: preclinical and phase 1 data. ESC Heart Fail. 2021;8(1):652-661.
- 2. WAINUA®
(eplontersen) [prescribing information]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2025. - 3. AMVUTTRA™ (vutrisiran) [prescribing information]. Cambridge, MA: Alnylam Pharmaceuticals, Inc.; March 2025.
- 4. ONPATTRO® (patisiran) [prescribing information]. San Diego, CA: Alnylam Pharmaceuticals, Inc; January 2023.
- 5. TEGSEDI® (inotersen) [prescribing information]. Waltham, MA; Sobi Inc;
January 2024. - 6. Crooke ST, Baker BF, Xia S, et al. Integrated assessment of the clinical performance of GalNAc3-conjugated 2'-O-methoxyethyl chimeric antisense oligonucleotides: I. Human volunteer experience. Nucleic Acid Ther. 2019;29(1):16-32.
IMPORTANT SAFETY INFORMATION
WARNINGS AND PRECAUTIONS
- Reduced Serum Vitamin A Levels and Recommended Supplementation WAINUA leads to a decrease in serum vitamin A levels. Supplement with recommended daily allowance of vitamin A. Refer patient to an ophthalmologist if ocular symptoms suggestive of vitamin A deficiency occur.
ADVERSE REACTIONS
Most common adverse reactions (≥9% in WAINUA-treated patients) were vitamin A decreased (15%) and vomiting (9%).
INDICATION
WAINUA injection, for subcutaneous use, 45 mg is indicated for the treatment of the polyneuropathy of hereditary transthyretin-mediated amyloidosis
in adults.


