HOW TO DIAGNOSE
Understand the key symptoms that raise clinical suspicion of polyneuropathy1
hATTR-PN is a rapidly progressive and fatal disease that requires urgent identification2,3
Identify patients with undiagnosed hATTR-PN by recognizing the signs and symptoms
NEUROLOGIC EVALUATION IN AMYLOIDOSIS1
Autonomic dysfunction
- Orthostatic hypotension
- Diarrhea/constipation
- Urinary retention
- Erectile dysfunction
Sensory symptoms
- Numbness/pain in feet
- Inability to sense pain/temperature
- Impaired balance/falls
Motor loss
- Tripping, foot drop
- Difficulty with stairs
- Usually after sensory symptoms
Physical examination
- Muscle weakness
- Distal sensory loss
- Reduced or absent reflexes
- Wide-based unsteady gait
Patients experienced symptom onset between 2.5 to 10 years prior to diagnosis9-11
The scales below can be helpful tools for assessing and documenting the severity of polyneuropathy impairment in your patients with hATTR-PN2
| Score | Polyneuropathy disability (PND) description |
|---|---|
| I | Sensory disturbances in extremities; preserved walking capability |
| II | Difficulty walking but no need for a stick or crutches |
| IIIa | Requires 1 stick or crutch for walking |
| IIIb | Requires 2 sticks or crutches for walking |
| IV | Confined to a wheelchair or bed |
| Stage | Familial amyloid polyneuropathy (FAP) description |
|---|---|
| I | Neuropathy limited to the lower limbs; walking without help |
| II | Progression of neuropathy in lower limbs; needs assistance when walking; muscles of the hands becoming weak |
| III | Confined to a wheelchair or bed; generalized weakness and areflexia |
Patients experienced symptom onset between 2.5 to 10 years prior to diagnosis9-11
Diagnostic assessment tool for identifying patients with hATTR-PN
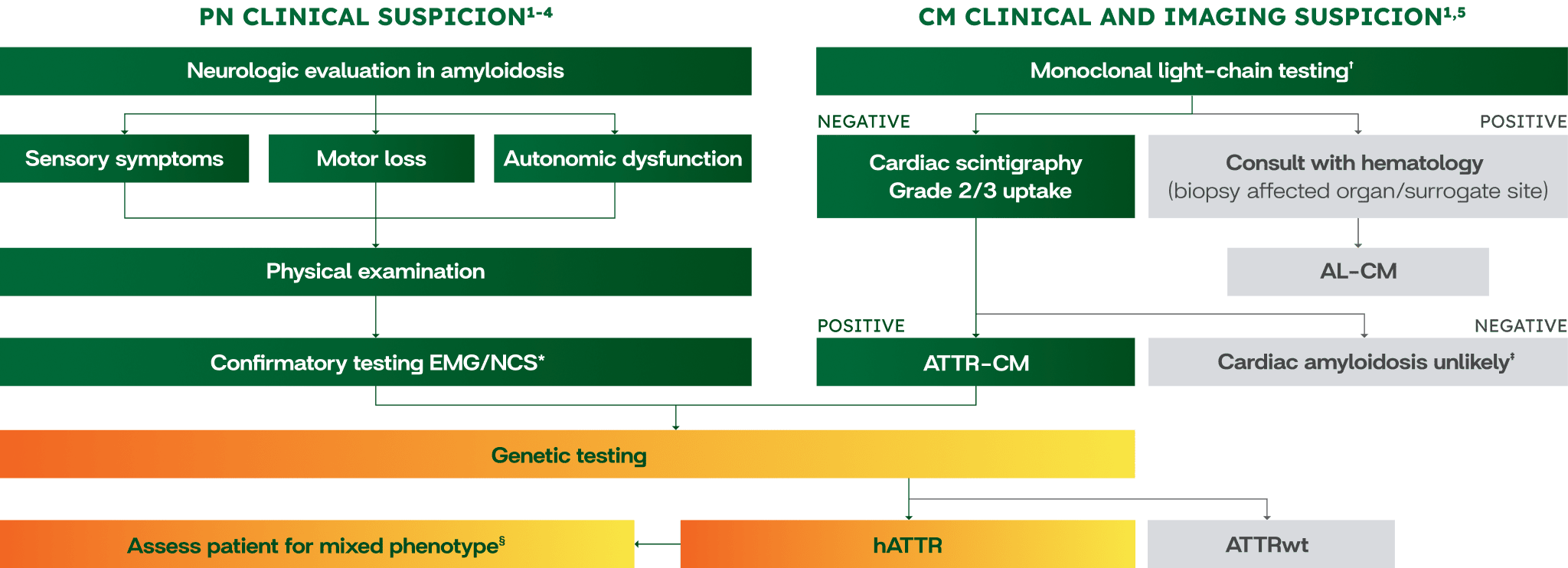

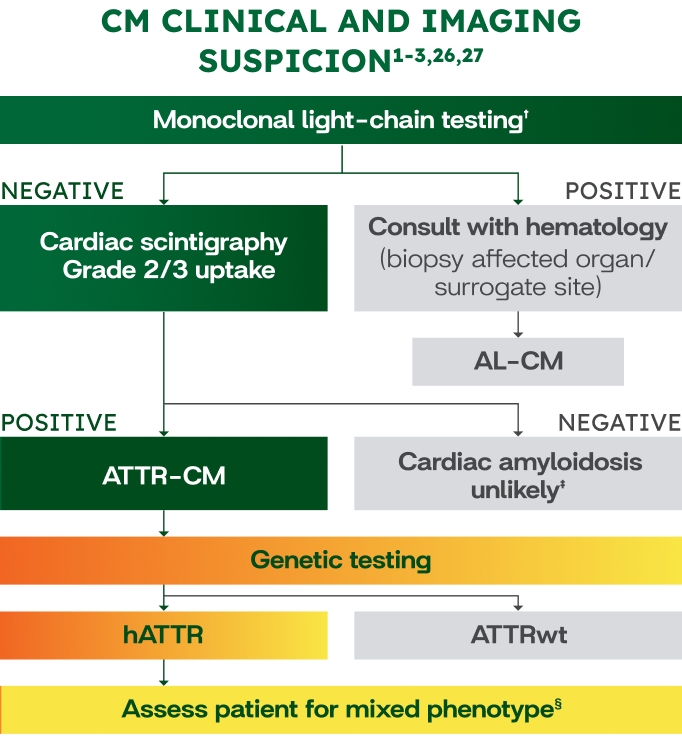
Algorithms are for illustrative purposes only.
Expert consensus and clinical guidelines:
Recommend genetic testing for all patients with ATTR1-8
WAINUA is not indicated for the treatment of cardiomyopathy symptoms.
- *Small-fiber neuropathy is not detected by conventional NCSs; therefore, a skin biopsy may be performed.1
- †Monoclonal protein screen should be performed to rule out AL-CM with sFLC, SIFE, and UIFE.1
- ‡Consider biopsy if cardiac scintigraphy is negative or equivocal and clinical suspicion is high.1
- §Including peripheral neuropathy (numbness, paresthesia, imbalance) or autonomic dysfunction (orthostatic hypotension, GI symptoms).1
Patients experienced symptom onset between 2.5 to 10 years prior to diagnosis9-11
Recognize the pattern of hATTR over time
See an illustrative case that may reflect your patient's experience
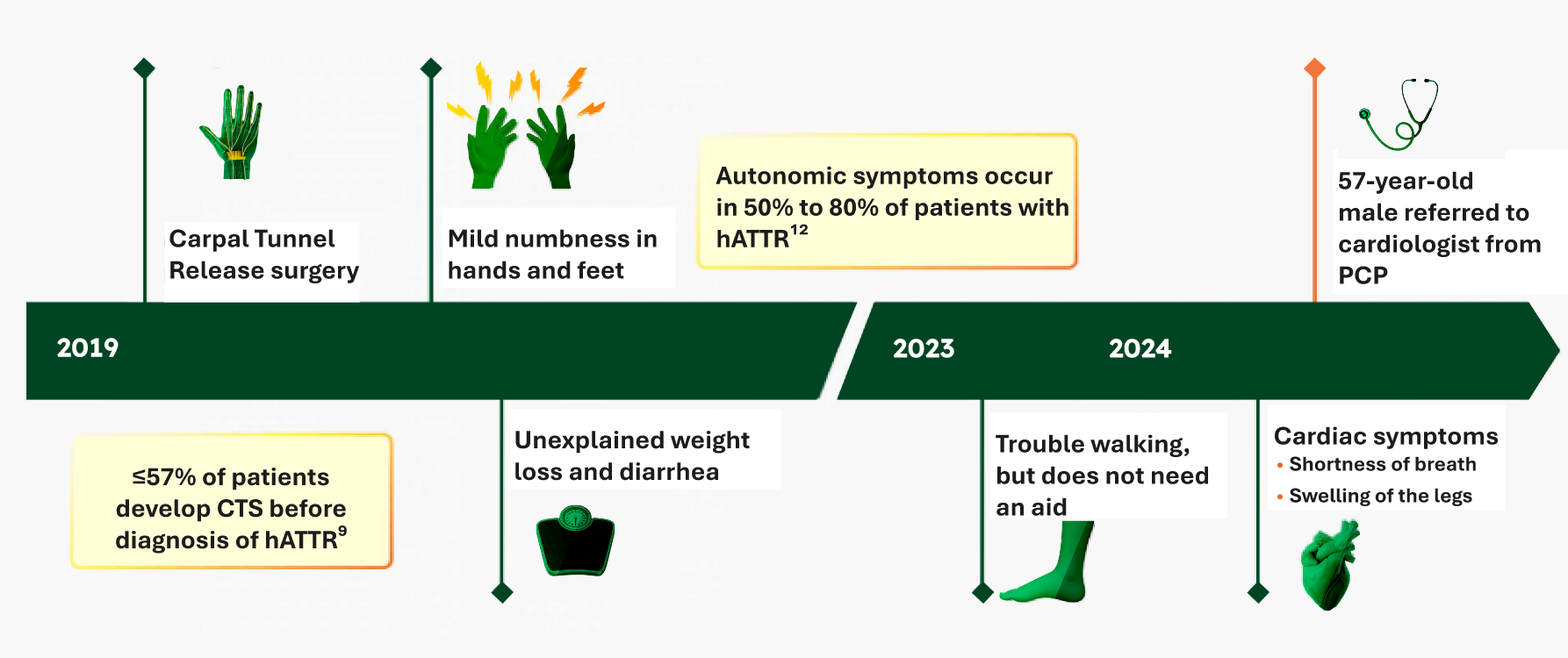
Suspicion of hATTR with mixed phenotype Further assessment and testing needed1-5
WAINUA is not indicated for the treatment of cardiomyopathy symptoms
Consider genetic testing to rule out hATTR-PN
- *Presence and severity of specific symptoms may depend on the duration and control of diabetes.13
- †CIDP may present with various phenotypes. This chart describes characteristics of atypical CIDP.19,21
- ‡Symptoms of motor involvement become more prominent as hATTR-PN progresses.12
- §GI manifestations, such as chronic diarrhea and constipation, are often considered autonomic symptoms.3,12
- ||Over 50% of patients develop CTS before diagnosis.15
- ¶Polyneuropathy due to ATTR can be 10 times more rapid than diabetic neuropathy.29
See how WAINUA targets TTR at
the source
Learn how WAINUA can affect
serum TTR levels
FOOTNOTES
Afib, atrial fibrillation; AL-CM, light-chain cardiac amyloidosis; ATTR, transthyretin-mediated amyloidosis; ATTR-CM, cardiomyopathy of transthyretin-mediated amyloidosis; ATTRwt, wild-type transthyretin-mediated amyloidosis; CIDP, chronic inflammatory demyelinating polyneuropathy; CTS, carpal tunnel syndrome; EMG, electromyography; GI, gastrointestinal; hATTR, hereditary transthyretin-mediated amyloidosis; hATTR-PN, polyneuropathy of hereditary transthyretin-mediated amyloidosis; HFpEF, heart failure with preserved ejection fraction; NCS, nerve conduction study; PCP, primary care provider; PN, polyneuropathy; sFLC, serum free light chain; SIFE, serum immunofixation electrophoresis; SNAP, sensory nerve action potential; TTR, transthyretin; UIFE, urine immunofixation electrophoresis.
REFERENCES
- 1. Kittleson MM, Ruberg FL, Ambardekar AV, et al; Writing Committee. 2023 ACC Expert Consensus Decision Pathway on comprehensive multidisciplinary care for the patient with cardiac amyloidosis: a report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol. 2023;81(11):1076-1126.
- 2. Adams D, Ando Y, Beirão JM, et al. Expert consensus recommendations to improve diagnosis of ATTR amyloidosis with polyneuropathy. J Neurol. 2021;268(6):2109-2122.
- 3. Gertz M, Adams D, Ando Y, et al. Avoiding misdiagnosis: expert consensus recommendations for the suspicion and diagnosis of transthyretin amyloidosis for the general practitioner. BMC Fam Pract. 2020;21(1):198.
- 4. Alcantara M, Mezei MM, Baker SK, et al. Canadian guidelines for hereditary transthyretin amyloidosis polyneuropathy management. Can J Neurol Sci. 2022;49(1):7-18.
- 5. Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145(18):e895-e1032.
- 6. Fine NM, Davis MK, Anderson K, et al. Canadian Cardiovascular Society/Canadian Heart Failure Society joint position statement on the evaluation and management of patients with cardiac amyloidosis. Can J Cardiol. 2020;36(3):322-334.
- 7. Brito D, Albrecht FC, de Arenaza DP, et al. World Heart Federation consensus on transthyretin amyloidosis cardiomyopathy (ATTR-CM). Glob Heart. 2023;18(1):59.
- 8. Arbelo E, Protonotarios A, Gimeno JR, et al; ESC Scientific Document Group. 2023 ESC Guidelines for the management of cardiomyopathies. Eur Heart J. 2023;44(37):3503-3626.
- 9. Aus dem Siepen F, Hein S, Prestel S, et al. Carpal tunnel syndrome and spinal canal stenosis: harbingers of transthyretin amyloid cardiomyopathy? Clin Res Cardiol. 2019;108(12):1324-1330.
- 10. Cappelli F, Zampieri M, Fumagalli C, et al. Tenosynovial complications identify TTR cardiac amyloidosis among patients with hypertrophic cardiomyopathy phenotype. J Intern Med. 2021;289(6):831-839.
- 11. Barroso FA, Coelho T, Dispenzieri A, et al; THAOS Investigators. Characteristics of patients with autonomic dysfunction in the Transthyretin Amyloidosis Outcomes Survey (THAOS). Amyloid. 2022;29(3):175-183.
- 12. Nativi-Nicolau JN, Karam C, Khella S, Maurer MS. Screening for ATTR amyloidosis in the clinic: overlapping disorders, misdiagnosis, and multiorgan awareness. Heart Fail Rev. 2022;27(3):785-793.
- 13. Feldman EL, Callaghan BC, Pop-Busui R, et al. Diabetic neuropathy. Nat Rev Dis Primers. 2019;5(1):42.
- 14. Waddington-Cruz M, Wixner J, Amass L, Kiszko J, Chapman D, Ando Y; THAOS Investigators. Characteristics of patients with late- vs. early-onset Val30Met transthyretin amyloidosis from the Transthyretin Amyloidosis Outcomes Survey (THAOS). Neurol Ther. 2021;10(2):753-766.
- 15. Karam C, Dimitrova D, Christ M, Heitner SB. Carpal tunnel syndrome and associated symptoms as first manifestation of hATTR amyloidosis. Neurol Clin Pract. 2019;9(4):309-313.
- 16. Poli L, Labella B, Cotti Piccinelli S, et al. Hereditary transthyretin amyloidosis: a comprehensive review with a focus on peripheral neuropathy. Front Neurol. 2023;14:1242815.
- 17. Lin X, Yarlas A, Vera-Llonch M, et al. Rate of neuropathic progression in hereditary transthyretin amyloidosis with polyneuropathy and other peripheral neuropathies: a systematic review and meta-analysis. BMC Neurol. 2021;21(1):70.
- 18. Adams D, Coelho T, Obici L, et al. Rapid progression of familial amyloidotic polyneuropathy: a multinational natural history study. Neurology. 2015;85(8):675-682.
- 19. Lewis RA, van Doorn PA, Sommer C. Tips in navigating the diagnostic complexities of chronic inflammatory demyelinating polyradiculoneuropathy. J Neurol Sci. 2022;443:120478.
- 20. Zis P, Sarrigiannis PG, Rao DG, Hewamadduma C, Hadjivassiliou M. Chronic idiopathic axonal polyneuropathy: a systematic review. J Neurol. 2016;263(10):1903-1910.
- 21. Fisse AL, Motte J, Grüter T, Sgodzai M, Pitarokoili K, Gold R. Comprehensive approaches for diagnosis, monitoring and treatment of chronic inflammatory demyelinating polyneuropathy. Neurol Res Pract. 2020;2:42.
- 22. Vrancken AFJE, Notermans NC, Wokke JHJ, Franssen H. The realistic yield of lower leg SNAP amplitudes and SRAR in the routine evaluation of chronic axonal polyneuropathies. J Neurol. 2008;255(8):1127-1135.
- 23. Lehmann HC, Wunderlich G, Fink GR, Sommer C. Diagnosis of peripheral neuropathy. Neurol Res Pract. 2020;2:20.
- 24. Natesan V, Kim SJ. Diabetic nephropathy - a review of risk factors, progression, mechanism, and dietary management. Biomol Ther (Seoul). 2021;29(4):365-372.
- 25. Bansal V, Kalita J, Misra UK. Diabetic neuropathy. Postgrad Med J. 2006;82(964):95-100.
- 26. Van den Bergh PYK, van Doorn PA, Hadden RDM, et al. European Academy of Neurology/Peripheral Nerve Society guideline on diagnosis and treatment of chronic inflammatory demyelinating polyradiculoneuropathy: report of a joint task force-second revision. J Peripher Nerv Syst. 2021;26(3):242-268.
- 27. Visser NA, Vrancken AFJE, van der Schouw YT, van den Berg LH, Notermans NC. Chronic idiopathic axonal polyneuropathy is associated with the metabolic syndrome. Diabetes Care. 2013;36(4):817-822.
- 28. Visser NA, Notermans NC, Degen LAR, de Kruijk JR, van den Berg LH, Vrancken AFJE. Chronic idiopathic axonal polyneuropathy and vitamin B6: a controlled population-based study. J Peripher Nerv Syst. 2014;19(2):136-144.
- 29. Berk JL, Suhr OB, Obici L, et al. Repurposing diflunisal for familial amyloid polyneuropathy: a randomized clinical trial. JAMA. 2013;310(24):2658-2667.
IMPORTANT SAFETY INFORMATION
WARNINGS AND PRECAUTIONS
- Reduced Serum Vitamin A Levels and Recommended Supplementation WAINUA leads to a decrease in serum vitamin A levels. Supplement with recommended daily allowance of vitamin A. Refer patient to an ophthalmologist if ocular symptoms suggestive of vitamin A deficiency occur.
ADVERSE REACTIONS
Most common adverse reactions (≥9% in WAINUA-treated patients) were vitamin A decreased (15%) and vomiting (9%).
INDICATION
WAINUA injection, for subcutaneous use, 45 mg is indicated for the treatment of the polyneuropathy of hereditary transthyretin-mediated amyloidosis in adults.



