HER2 can be overexpressed in many solid tumors beyond gastric and breast1
That’s why HER2 IHC testing is recommended across multiple tumor types.2-12 Your role in evaluating the HER2 biomarker further is critical for patient care. We cannot over-express this enough.
The clinical relevance of HER2 positivity (IHC 3+)
- HER2 positivity has long been associated with a poor prognosis, including increased recurrence and decreased outcomes across different tumor types13-18
- HER2 is broadly expressed and clinically relevant across solid tumors, including NSCLC, endometrial, cervical, ovarian, bladder, biliary tract, CRC, pancreatic, gastric, and breast cancers18,19
Prevalence of HER2 alterations across solid tumorsa
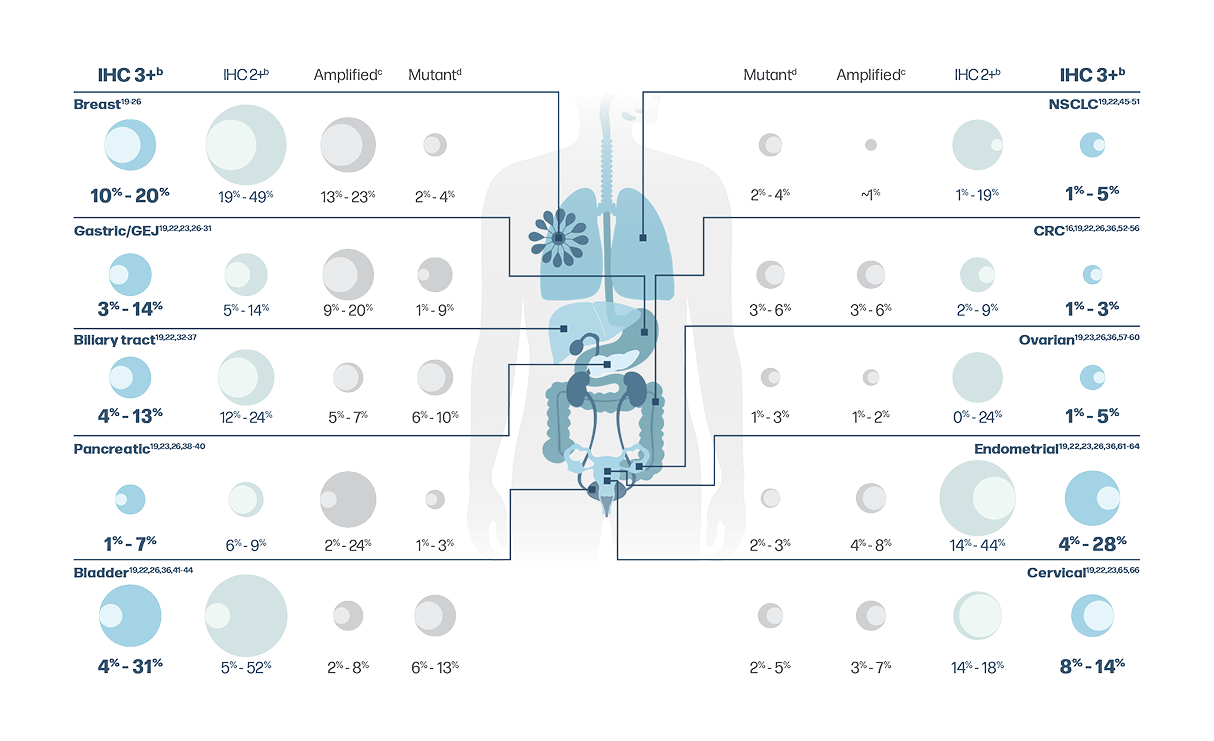
aIndividual tumor prevalence numbers reflect US and ex-US populations. Due to limited testing of IHC in the US, data from a global population have been included.
bHER2 protein expression is measured using IHC with confirmatory ISH testing in cases of equivocal results.67,68
cHER2 (ERBB2) amplification is measured using ISH techniques, including FISH, CISH, and SISH, or by NGS.50,69
dHER2 (ERBB2) mutations are detected through sequencing techniques, such as NGS.50,54
HER2 protein overexpression
(IHC 3+) is distinct from genomic HER2 (ERBB2) alterations
Overview of HER2 overexpression, gene amplification, and gene
mutation67,68,70,71
| Alteration | HER2 protein overexpression |
HER2 (ERBB2) gene amplification |
HER2 (ERBB2) gene mutation |
|---|---|---|---|
| Definition | Overabundance of HER2 protein on the surface of tumor cells |
Elevated |
Activating |
| Testing method | IHC | ISH or NGSe | NGSd |
- There are 3 distinct types of HER2 alteration that may lead to oncogenic activation of the
HER2 pathway: HER2 protein
overexpression, HER2 (ERBB2) gene
amplification, and HER2 (ERBB2) gene
mutation67,68,70,71 - NGS, FISH, and liquid biopsy do not detect HER2 protein overexpression (IHC 3+)68,70
eISH is a molecular testing method that detects
HER2 DNA copy number, while NGS detects both
HER2 DNA copy number and/or HER2
mutations.70,71
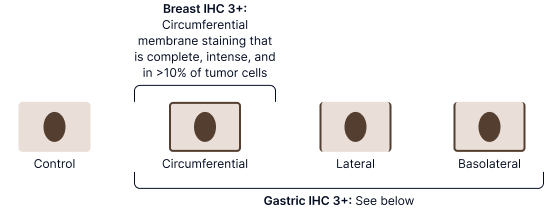
HER2 IHC scoring for solid tumor types other than breast cancer
- HER2 IHC testing is well established in gastric cancer, and the gastric scoring criteria have been applied to other solid tumor types, including NSCLC, colorectal, and gynecologic tumors2,7,68,73
- ASCO-CAP gastric cancer scoring criteria allow for more staining heterogeneity than ASCO-CAP breast cancer scoring criteria67,68,74
- For example, gastric cancer scoring criteria define HER2 overexpression using basolateral and lateral membrane staining in addition to circumferential staining
Staining patterns required for IHC 3+ with each set of ASCO-CAP scoring criteria67,68
ASCO-CAP gastric cancer scoring criteria algorithm68,75
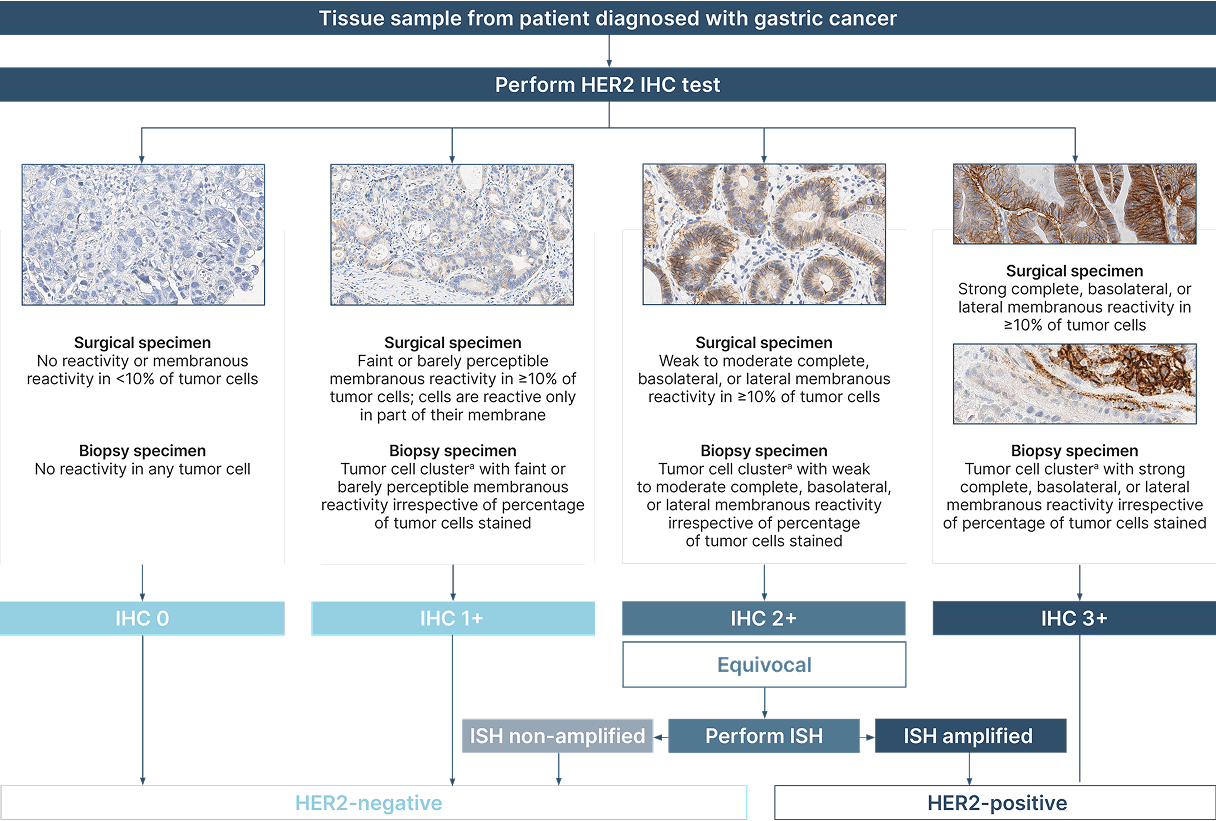
aTumor cell cluster (≥5 neoplastic cells).68
Figure adapted from: Bartley AN, et al. J Clin Oncol. 2017.68
Images provided by Discovery Life Sciences, Inc.
HER2 IHC testing recommendations
There are several key considerations in HER2 assay validation beyond the testing guidance for breast and gastric cancers, including which assay scoring system is used76
- ASCO-CAP gastric cancer scoring criteria may be applied in multiple solid tumor types to determine HER2 IHC status2,68,73,77
HER2 testing guidance and reporting tools across tumor types
| Pathology specialty | Metastatic/advanced tumor type | NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) testing guidance | CAP Biomarker Reporting Template for HER2 testing results | Applicable HER2 IHC scoring criteria |
|---|---|---|---|---|
| Breast | Breast | IHC, ISH3,67,78 | Breast Biomarker Reporting Template | ASCO–CAP breast scoring criteriaa |
| Gastrointestinal | Biliary tract | IHC, FISH, NGS69,79 | General IHC Quantitative Biomarkers Template | No universal guideline (ASCO–CAP gastric scoring criteria may be used) |
| Gastric/GEJ | IHC, ISH, NGS5,68,80,81 | Gastric HER2 Biomarker Reporting Template | ASCO–CAP gastric scoring criteriaa | |
| Pancreatic | IHC, NGS (amplification)6,79 | General IHC Quantitative Biomarkers Template | No universal guideline (ASCO–CAP gastric scoring criteria may be used) | |
| Colorectal | IHC, FISH, NGS7,8,77 | Colon and Rectum Biomarker Reporting Template | ASCO–CAP gastric scoring criteria, ASCO–CAP breast scoring criteria, or HERACLES trial scoring criteriab,c |
|
| Thoracic | NSCLC | IHC, NGS (mutation), targeted PCR2,82 | Lung Biomarker Reporting Template | No universal guideline (ASCO–CAP gastric scoring criteria may be used)d |
| Genitourinary | Bladder | IHC9,79 | General IHC Quantitative Biomarkers Template | No universal guideline (ASCO–CAP gastric scoring criteria may be used) |
| Gynecologic | Endometrial | IHC, FISH10,68,73 | Gynecologic Biomarker Reporting Template | ASCO–CAP gastric scoring criteria or Fader trial guidelinesc |
| Ovarian | IHC11,68,73 | Gynecologic Biomarker Reporting Template | ASCO–CAP gastric scoring criteriac | |
| Cervical | IHC, FISH12,68,73 | Gynecologic Biomarker Reporting Template | ASCO–CAP gastric scoring criteriac |
aCriteria recommended by ASCO-CAP.
bThere is no consensus on whether breast or gastric cancer scoring criteria should be applied to colorectal cancer. Some clinical trials have utilized ASCO-CAP breast scoring criteria, and others have utilized ASCO-CAP gastric cancer scoring criteria. Laboratory directors are asked to exercise their discretion as to which scoring criteria to apply.76
cCriteria included in CAP reporting template.
dScoring criteria for anti-HER2 monoclonal antibody/antibody-drug conjugate therapy is currently based on criteria initially applied in gastric carcinoma, which utilizes different scoring criteria than that utilized for breast carcinoma and employs different scoring criteria for small biopsy specimens versus resections.2
HER2 IHC reporting across tumor types
 Breast
Breast Colon and rectum
Colon and rectum Lung
Lung Gynecologic
Gynecologic Gastric HER2
Gastric HER2 Head and neck
Head and neckFor other tumor types, the CAP General IHC Quantitative Biomarkers Template is available.
Access all of the CAP templates listed above
Example of CAP validation guidance for IHC assays in solid tumors outside of breast and gastric cancersa

Extending a previously validated assay to colorectal cancer76
- If the laboratory director intends to perform HER2 IHC on colorectal tumors using a previously validated assay and scoring criteria combination, the laboratory director has the discretion to extend the initial validation to colorectal cancer by assessing a representative sample
of colorectal cancer

Validating a new assay in multiple tumor types76
- If a laboratory is initially validating a new HER2 assay and intends to use the same scoring criteria in breast and colorectal cancers, then both cancer types should be included in the set of 20 positive and 20 negative tissues constituting the validation
- It is not the intent of this recommendation that every assay, scoring criteria, and tumor type combination be subject to the requirement of 20 positive and
20 cases for each validation
- It is not the intent of this recommendation that every assay, scoring criteria, and tumor type combination be subject to the requirement of 20 positive and
aUpdated in 2024.
Case study examples (IHC 3+)75
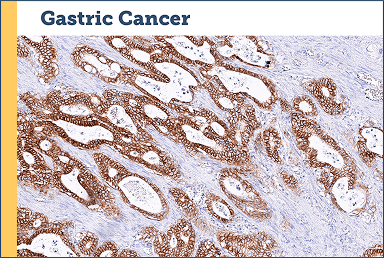
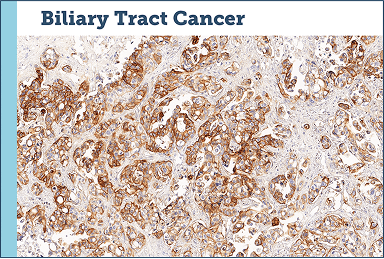
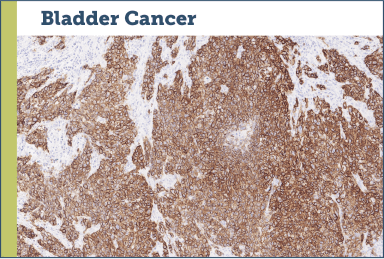
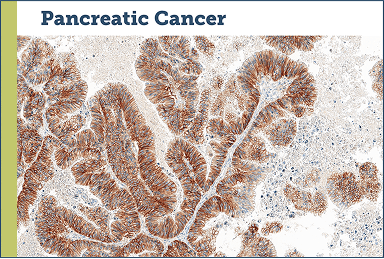
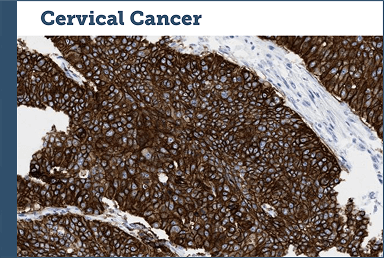
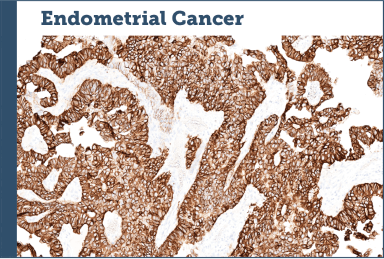
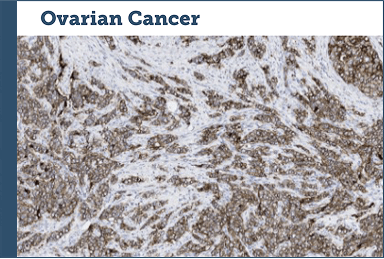
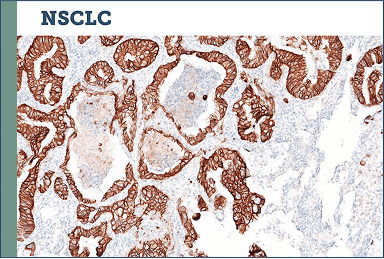
Images provided by Discovery Life Sciences, Inc.
HER2 COULD BE HERE, TOO
Perform HER2 IHC testing across metastatic solid tumor types
- HER2 IHC testing is recommended in NCCN Guidelines® across numerous solid tumor types2-12
- Consider using ASCO-CAP gastric cancer scoring criteria across solid tumor types, outside of breast2,68,73,77
- Due to the biology of the different alterations, NGS, FISH, and liquid biopsy do not detect HER2 protein overexpression (IHC 3+)68,70
- HER2 IHC testing should be performed, regardless of NGS or ISH results, to ensure patients are accurately identified68,70-72

Perform HER2
IHC testing across metastatic solid tumor types to inform patient care
ASCO, American Society of Clinical Oncology; CAP, College of American Pathologists; CISH, chromogenic in situ hybridization; CRC, colorectal cancer; DNA, deoxyribonucleic acid; ERBB2, erb-b2 receptor tyrosine kinase 2; FISH, fluorescence in situ hybridization; GEJ, gastroesophageal junction; HER2, human epidermal growth factor receptor 2; IHC, immunohistochemistry; ISH, in situ hybridization; mNSCLC, metastatic non-small cell lung cancer; NCCN, National Comprehensive Cancer Network; NGS, next-generation sequencing; NSCLC, non-small cell lung cancer; SISH, silver-enhanced in situ hybridization.
1. Yan M, et al. Cancer Metastasis Rev. 2015;34(1):157–164. 2. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Non-Small Cell Lung Cancer V.7.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed July 10, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 3. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Breast Cancer V.4.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed April 17, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 4. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Biliary Tract Cancers V.2.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed July 2, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 5. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Gastric Cancer V.2.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed April 4, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 6. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Pancreatic Adenocarcinoma V.2.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed February 3, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 7. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Colon Cancer V.4.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed June 27, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 8. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Rectal Cancer V.2.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed March 31, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 9. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Bladder Cancer V.1.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed March 25, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 10. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Uterine Neoplasms V.3.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed March 7, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 11. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Ovarian Cancer Including Fallopian Tube Cancer and Primary Peritoneal Cancer V.3.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed July 16, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 12. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Cervical Cancer V.4.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed March 24, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 13. Abrahao-Machado LF, et al. World J Gastroenterol. 2016;22(19):4619-4625. 14. Morrison C, et al. J Clin Oncol. 2006;24(15):2376-2385. 15. Luo H, et al. PLoS One. 2018;13(1):e0191972. 16. Wang XY, et al. World J Gastrointest Oncol. 2019;11(4):335-347. 17. Wang S, et al. Mod Pathol. 2023;36(6):100148. 18. Iqbal N, et al. Mol Biol Int. 2014;2014:852748. 19. Uzunparmak B, et al. Ann Oncol. 2023;34(11):1035-1046. 20. Farshid G, et al. Mod Pathol. 2020;33(9):1783-1790. 21. Rakha EA, et al. Br J Cancer. 2021;124(11):1836-1842. 22. Connell CM, et al. ESMO Open. 2017;2(5):e000279. 23. Chmielecki J, et al. Oncologist. 2015;20(1):7-12. 24. Owens MA, et al. Clin Breast Cancer. 2004;5(1):63-69. 25. Bose R, et al. Cancer Discov. 2013;3(2):224-237. 26. Subramanian J, et al. Oncologist. 2019;24(12):e1303-e1314. 27. Subasinghe D, et al. J Int Med Res. 2023;51(2):3000605231154403. 28. Van Cutsem E, et al. Gastric Cancer. 2015;18(3):476-484. 29. Gao X, et al. Int J Surg. 2023;109(5):1330-1341. 30. Im SA, et al. Diagn Mol Pathol. 2011;20(2):94-100. 31. Cappellesso R, et al. Hum Pathol. 2015;46(5):665-672. 32. Vivaldi C, et al. Oncologist. 2020;25(10):886-893. 33. Roa I, et al. Gastrointest Cancer Res. 2014;7(2):42-48. 34. Harder J, et al. World J Gastroenterol. 2009;15(36):4511-4517. 35. Nakazawa K, et al. J Pathol. 2005;206(3):356-365. 36. Dumbrava EEI, et al. JCO Precis Oncol. 2019;3:PO.18.00345. 37. Li M, et al. Nat Genet. 2014;46(8):872-876. 38. Han SH, et al. Diagnostics (Basel). 2021;11(4):653. 39. Chou A, et al. Genome Med. 2013;5(8):78. 40. Li X, et al. World J Surg Oncol. 2016;14:38. 41. Moktefi A, et al. Mod Pathol. 2018;31(8):1270-1281. 42. Gårdmark T, et al. BJU Int. 2005;95(7):982-986. 43. Fleischmann A, et al. Eur Urol. 2011;60(2):350-357. 44. Moustakas G, et al. J Int Med Res. 2020;48(1):300060519895847. 45. Zinner RG, et al. Lung Cancer. 2004;44(1):99-110. 46. Heinmöller P, et al. Clin Cancer Res. 2003;9(14):5238-5243. 47. Takenaka M, et al. Anticancer Res. 2011;31(12):4631-4636. 48. Nagasaka M, et al. Clin Lung Cancer. 2022;23(1):52-59. 49. Volckmar AL, et al. Int J Cancer. 2019;145(3):649-661. 50. Zhuo X, et al. J Cancer Res Clin Oncol. 2023;149(5):2029-2039. 51. Zhao J, et al. JCO Precis Oncol. 2020;4:411-425. 52. Ingold-Heppner B, et al. Br J Cancer. 2014;111(10):1977-1984. 53. Henry JT, et al. Chin Clin Oncol. 2019;8(5):49. 54. El-Deiry WS, et al. Cancer Biol Ther. 2015;16(12):1726-1737. 55. Seo AN, et al. PLoS One. 2014;9(5):e98528. 56. Raghav KPS, et al. Clin Cancer Res. 2024;30(2):260-262. 57. Tuefferd M, et al. PLoS One. 2007;2(11):e1138. 58. Pils D, et al. Br J Cancer. 2007;96(3):485-491. 59. Ersoy E, et al. Int J Gynecol Pathol. 2022;41(4):313-319. 60. Chung YW, et al. J Gynecol Oncol. 2019;30(5):e75. 61. Buza N, et al. Mod Pathol. 2013;26(12):1605-1612. 62. Vermij L, et al. Histopathology. 2021;79(4):533-543. 63. Semiz HS, et al. Turk Patoloji Derg. 2023;39(1):55-63. 64. Halle MK, et al. Br J Cancer. 2018;118(3):378-387. 65. Shi H, et al. J Pathol Clin Res. 2021;7(1):86-95. 66. Panek G, et al. Gin Onkol. 2007;5(4):218-235. 67. Wolff AC, et al. Arch Pathol Lab Med 2023;147(9):993-1000. 68. Bartley AN, et al. J Clin Oncol. 2017;35(4):446-464. 69. Hanna WM, et al. Mod Pathol. 2014;27(1):4-18. 70. Ren S, et al. ESMO Open. 2022;7(1):100395. 71. Morsberger L, et al. Cancer Cell Int. 2022;22(1):350. 72. Shayeb AM, et al. JCO Precis Oncol. 2023;7:e2200604. 73. College of American Pathologists. Template for Reporting Results of Biomarker Testing of Specimens from Patients with Carcinoma of Gynecologic Origin. Version: 1.2.0.0. 74. Rüschoff J, et al. Mod Pathol. 2012;25(5):637-650. 75. Data on file. Daiichi Sankyo, Inc. Basking Ridge, NJ. 76. Goldsmith JD, et al. Arch Pathol Lab Med. 2024;148(6):e111-e153. 77. College of American Pathologists. Template for Reporting Results of Biomarker Testing of Specimens from Patients with Carcinoma of the Colon and Rectum. Version: 1.3.0.0. 78. College of American Pathologists. Template for Reporting Results of Biomarker Testing of Specimens from Patients with Carcinoma of the Breast. Version: 1.6.0.0. 79. College of American Pathologists. Template for Reporting Results of Biomarker Testing of Specimens from Patients with Carcinoma. Version: 1.1.0.1. 80. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Esophageal and Esophagogastric Junction Cancers V.3.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed April 22, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 81. Reporting Template for Reporting Results of Biomarker Testing of Specimens from Patients with Adenocarcinoma of the Stomach or Gastroesophageal Junction. Version: 17-1001. 82. Reporting Template for Reporting Results of Biomarker Testing of Specimens from Patients with Non-small Cell Carcinoma of the Lung. Version: 2.0.1.1. 83. CAP. Head and Neck Biomarker Reporting Template. Version: 2.2.0.0.
Get into the details
IHC Atlas:
Whole slide images
Examine real-world staining patterns across 100+ clinical cases to support HER2 IHC scoring in your practice.
Explore the cases
HER2 IHC reporting updates for gynecologic cancers
Overview of the CAP reporting template update and guidance for IHC scoring.
Read the article
HER2 IHC testing guidelines for mNSCLC
Overview of HER2 testing recommendations from NCCN Guidelines.
Read the article
Other Solid
Tumors
Resources
Discover educational materials on HER2 testing in solid tumors.









