EFFICACY
WAINUA demonstrated the power to stop polyneuropathy progression in adults with hATTR-PN*1-3

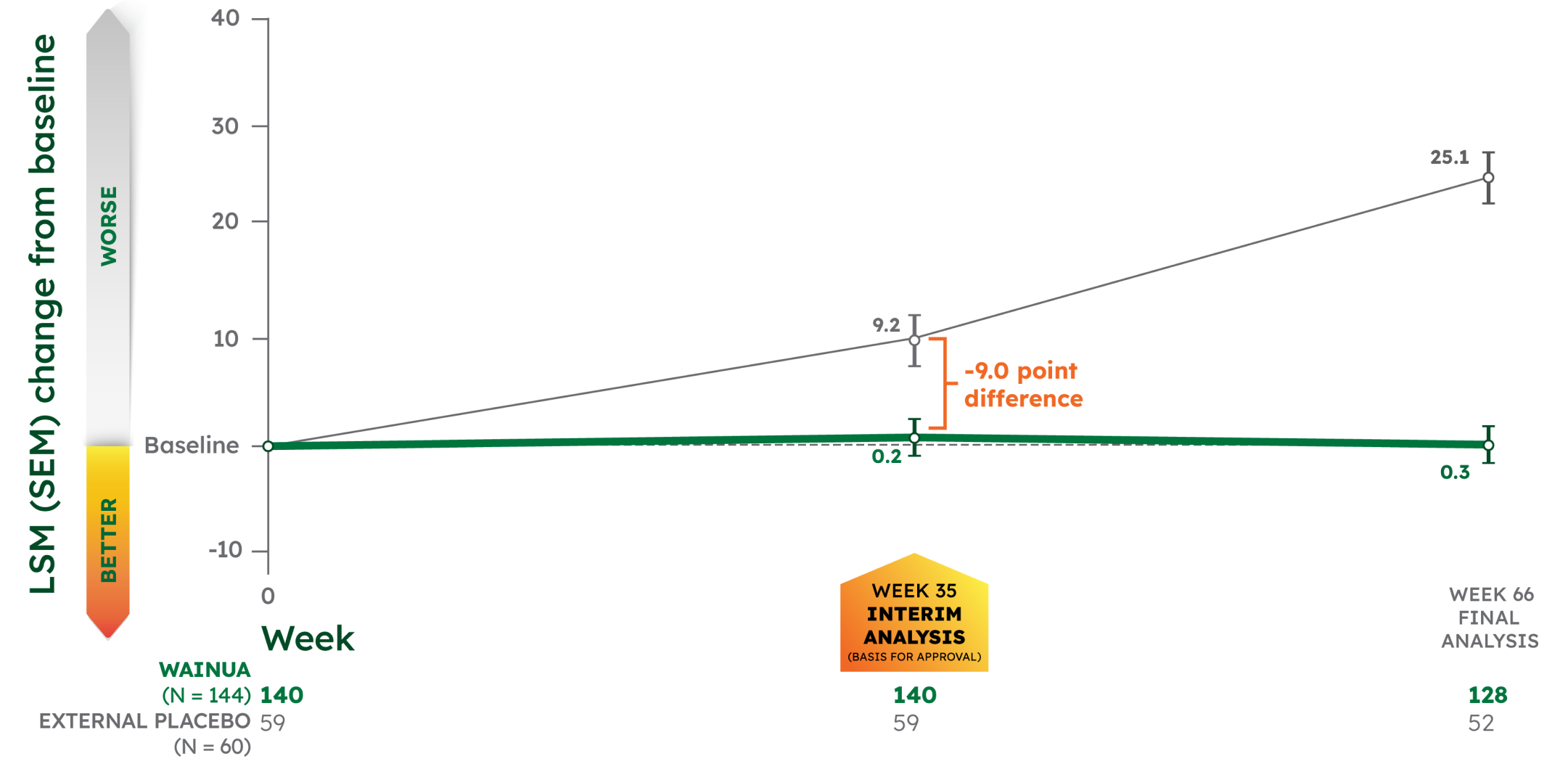
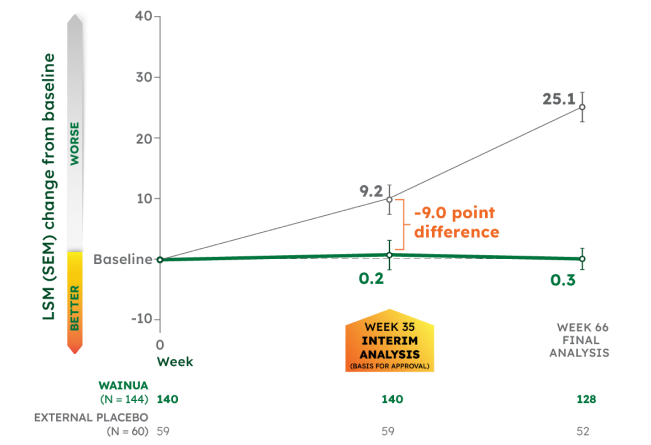
WAINUA significantly halted the progression of polyneuropathy impairment vs external placebo at Week 35 with sustained reductions through Week 661-3
LSM CHANGE FROM BASELINE IN mNIS+7 COMPOSITE SCORE†‡1-3
CO-PRIMARY ENDPOINT:
- -9.0 point significant difference with WAINUA vs placebo at Week 35 (95% CI, -13.5 to -4.5), P<0.001
- -24.8 point difference with WAINUA vs placebo at Week 66 (95% CI, -31.0 to -18.6)
NEURO-TTRansform was a phase 3, randomized, open-label trial. Data presented from baseline to Week 35 are from the interim analysis; data beyond Week 35 are from the final analysis. Because the primary and key secondary endpoints were statistically significant at Week 35, no further formal statistical analyses were performed at Week 65/66. See study design.1-4
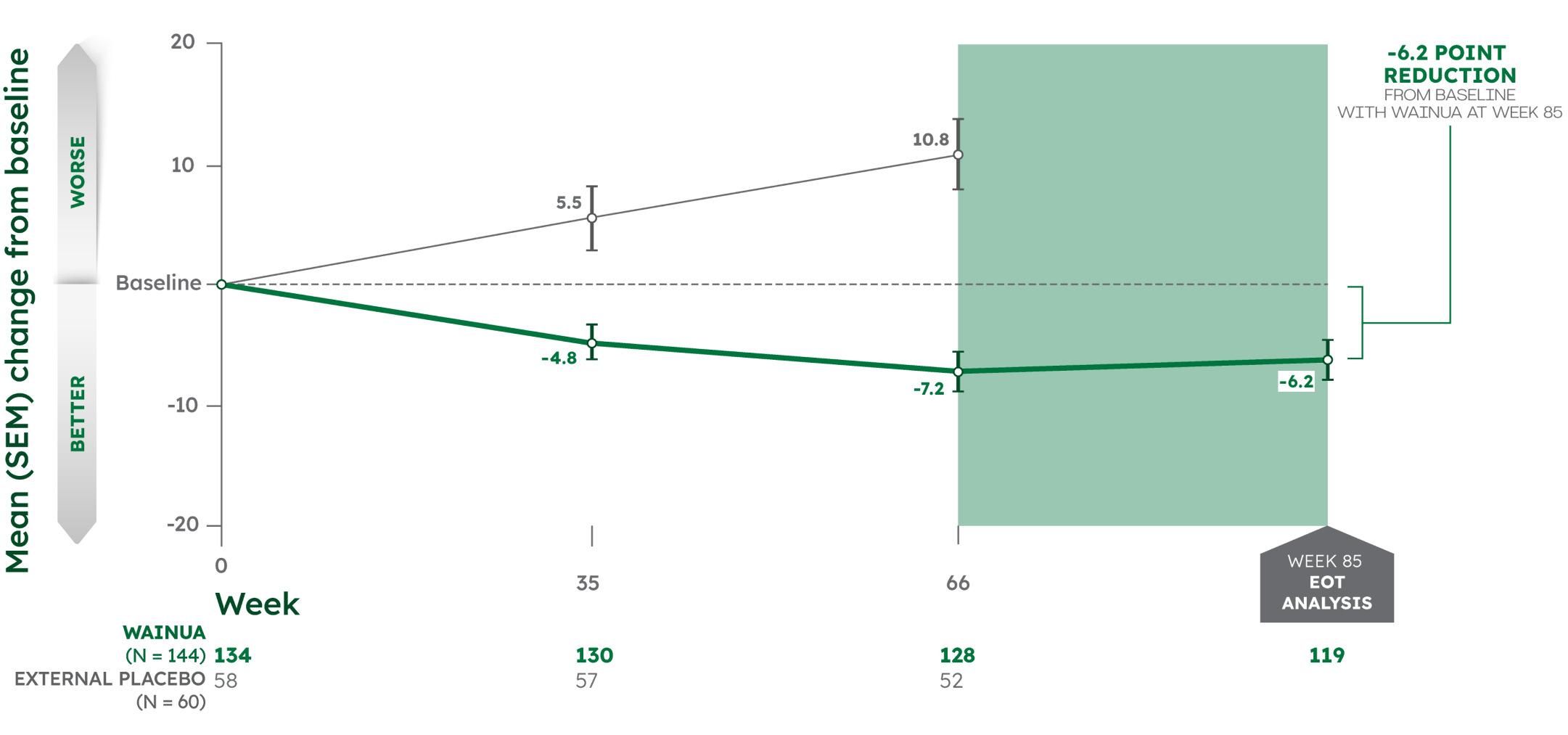
Effects on neuropathy impairment were sustained through Week 85 in adults with hATTR-PN2,3
Exploratory analysis
MEAN CHANGE FROM BASELINE IN mNIS+7 COMPOSITE SCORE*†2,3
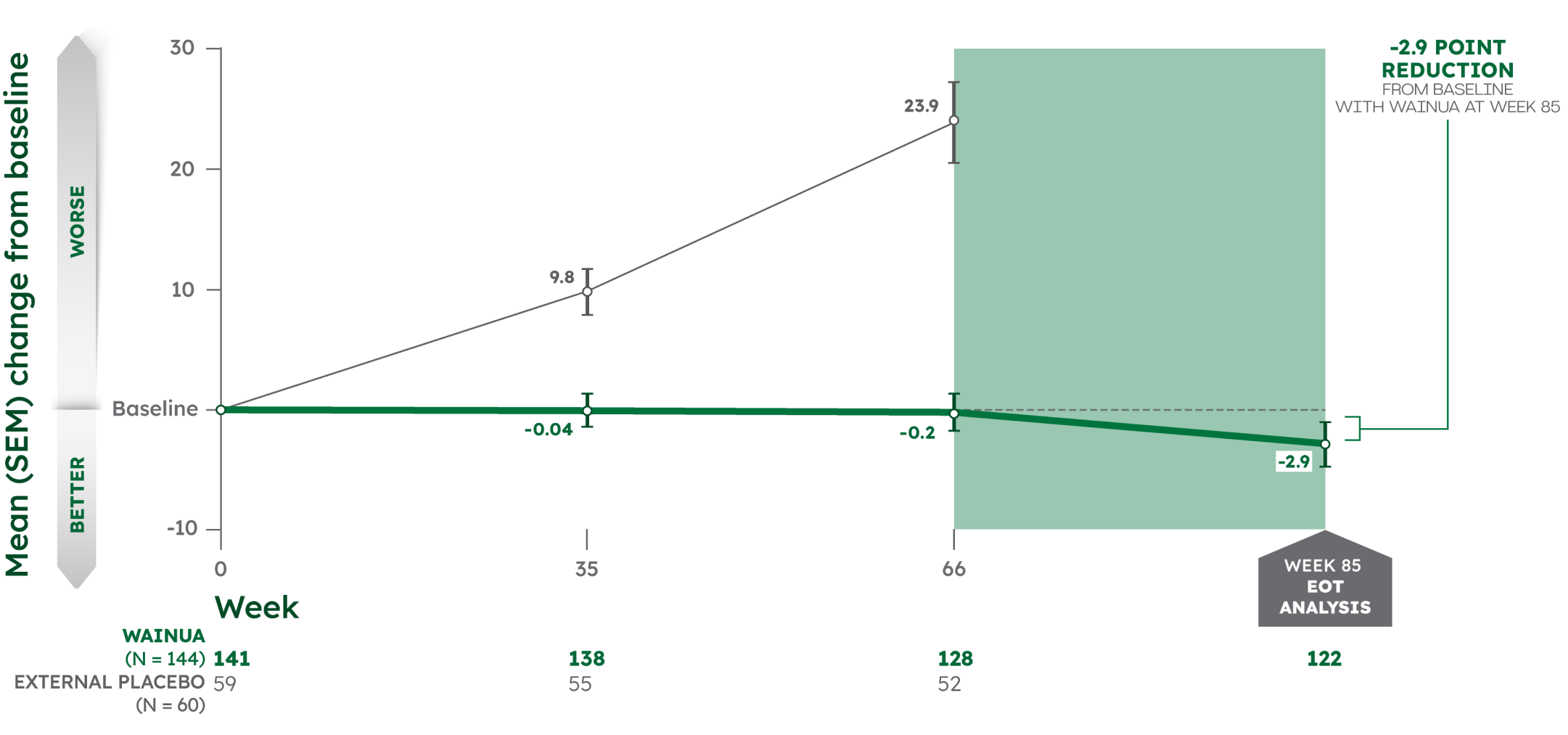
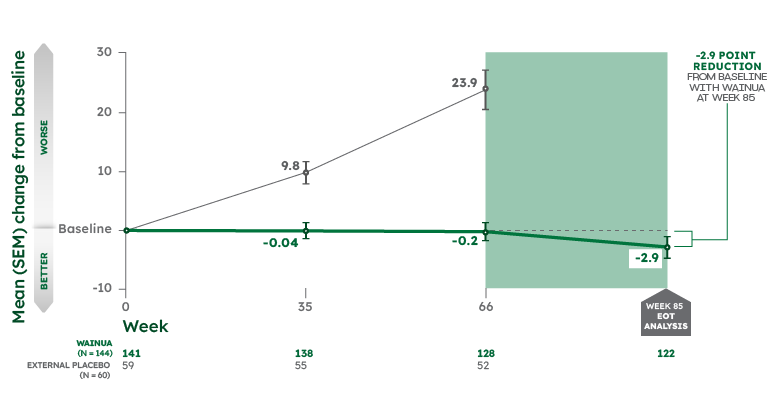
Note: From Week 66 to Week 85, there was no external placebo group because NEURO-TTR concluded at Week 66. Change in mNIS+7 composite score was assessed as an exploratory outcome at Week 85. No formal statistical analyses were performed at Week 85.3
mNIS+7 composite score is validated in hATTR-PN to quantify neurologic impairment and progression1,3,5
Scores measure muscle strength/weakness, quantitative sensory testing, sensations, reflexes, nerve conduction, and heart rate response to deep breathing.


MUSCLE WEAKNESS/STRENGTH
Assessed in 24 muscle groups
QUANTITATIVE SENSORY TESTING
(touch-pressure and heat-pain)
Assessed in up to 10 sites
SENSATIONS
Assessed touch, pressure, and pinprick
REFLEXES
Assessed in 5 muscle groups
NERVE CONDUCTION STUDIES
5 nerve assessments
HEART RATE RESPONSE
TO DEEP BREATHING
The higher the score, the greater the impairment
mNIS+7 composite score measures muscle weakness, sensation, and autonomic function1,3
*As measured by mNIS+7 composite score, validated score in hATTR-PN to quantify neurologic impairment and progression.1,3,5
†The validated version of the mNIS+7 score used in the trial had a range of -22.3 to 346.3 points, with higher scores representing a greater severity of disease.1
‡Improvement is defined as a negative change from baseline; patients with missing data were counted as not improved.3
Responder analysis for mNIS+7 composite score at Week 35 and Week 661-3
Responder analysis at Week 351
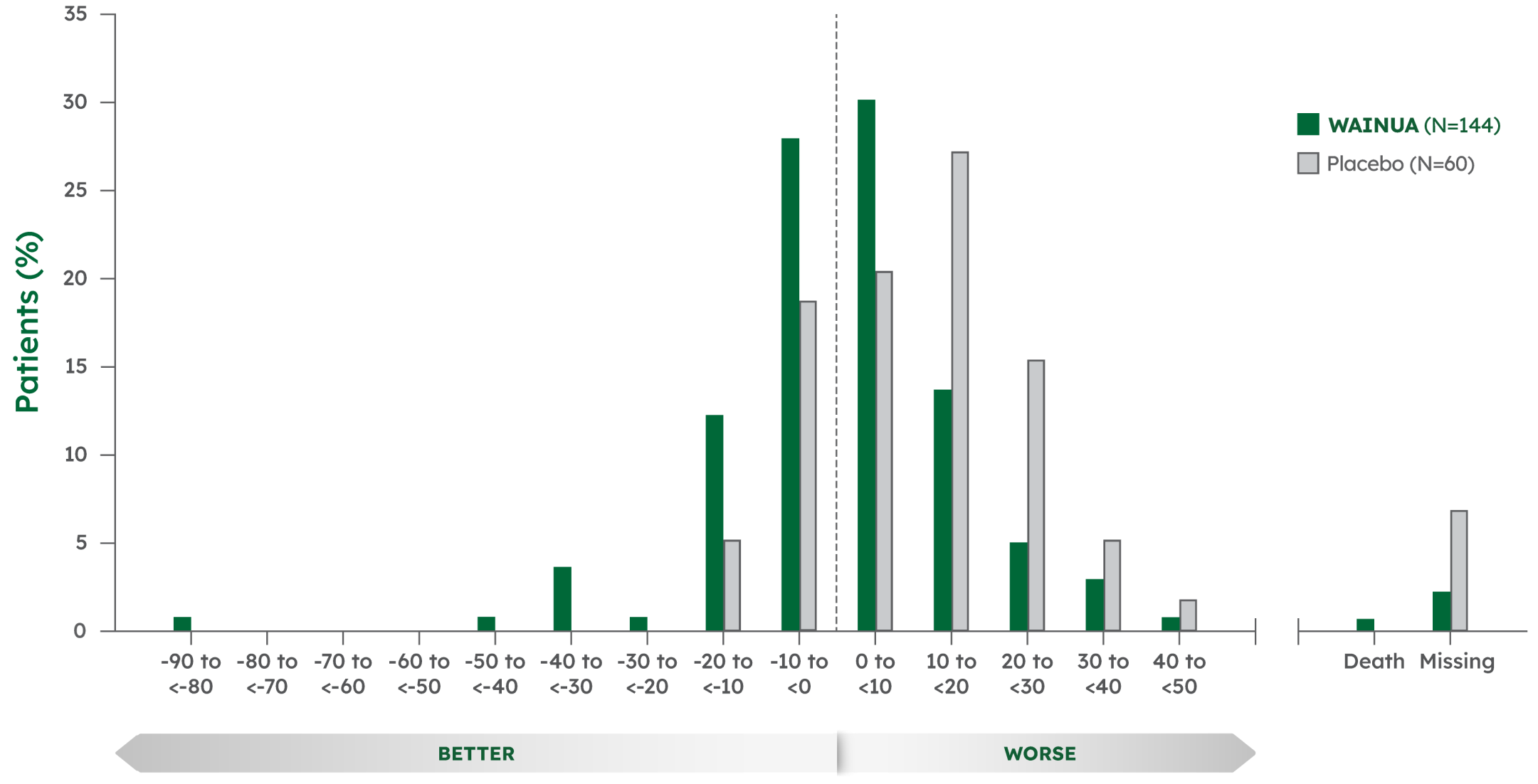
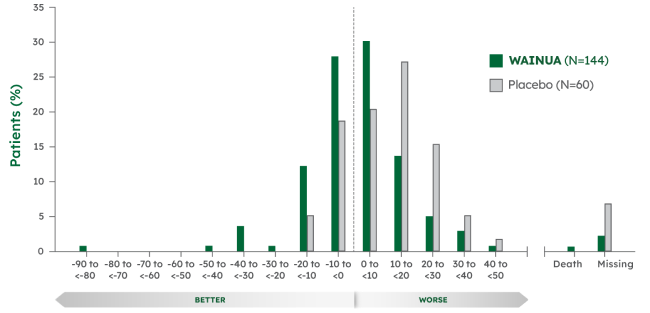
Patients treated with WAINUA had better mNIS+7 composite scores vs patients treated with placebo from baseline at Week 35 and Week 66.§2,3
- Week 35: WAINUA, 49% vs placebo, 24%
- Week 66 (post hoc analysis): WAINUA, 53% vs placebo, 19%
Note: A responder (improvement) was defined as a patient whose mNIS+7 composite score change from baseline was less than threshold value of 0.2,3
§At Week 35, the number of patients in each group of the full analysis set was used as the denominator (WAINUA, N=140; placebo, N=59).2
At Week 66, the number of patients with nonmissing values in each group of the full analysis set was used as the denominator (WAINUA, N=128; placebo, N=52).
WAINUA demonstrated the power to significantly and continuously improve quality of life in adults with hATTR-PN||1-3
WAINUA significantly improved quality of life vs external placebo at Week 35 with sustained effect through Week 661-3
LSM CHANGE FROM BASELINE IN NORFOLK QoL-DN TOTAL SCORE‡¶1-3
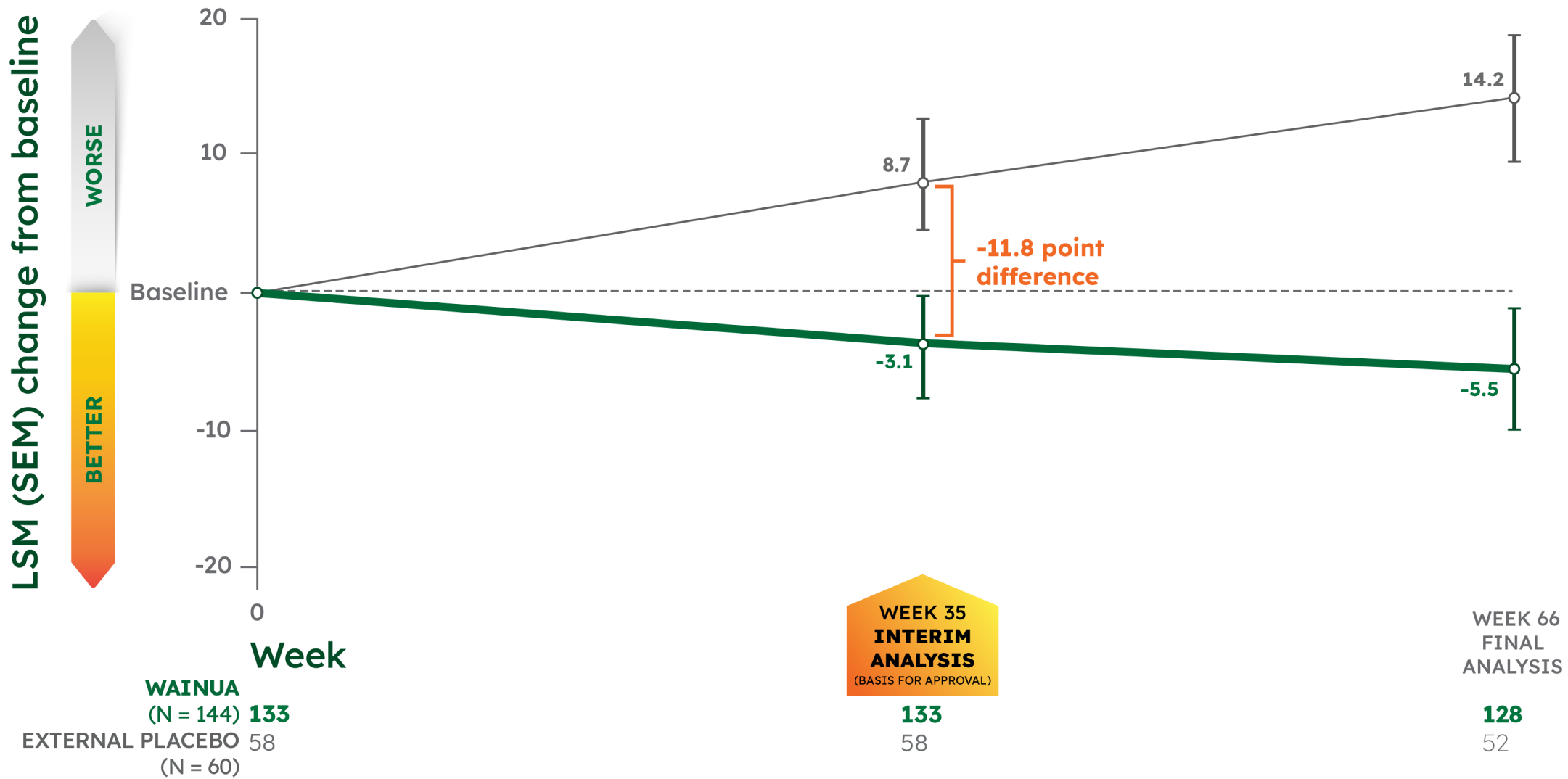
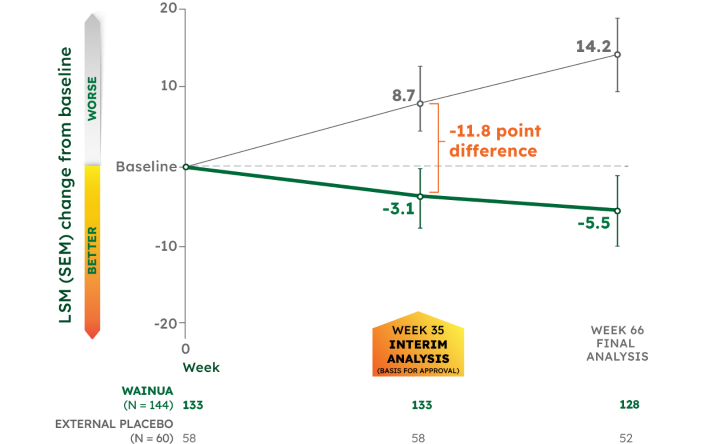
CO-PRIMARY ENDPOINT:
- -11.8 point significant difference with WAINUA vs placebo at Week 35 (95% CI, -16.8 to -6.8), P<0.001
- -19.7 point difference with WAINUA vs placebo at Week 66 (95% CI, -25.6 to -13.8)
NEURO-TTRansform was a phase 3, randomized, open-label trial. Data presented from baseline to Week 35 are from the interim analysis; data beyond Week 35 are from the final analysis. Because the primary and key secondary endpoints were statistically significant at Week 35, no further formal statistical analyses were performed at Week 65/66. See study design.1-4
Effects on quality of life were sustained through Week 85 in adults with hATTR-PN2,3
Exploratory analysis
MEAN CHANGE FROM BASELINE IN NORFOLK QoL-DN TOTAL SCORE‡¶2,3
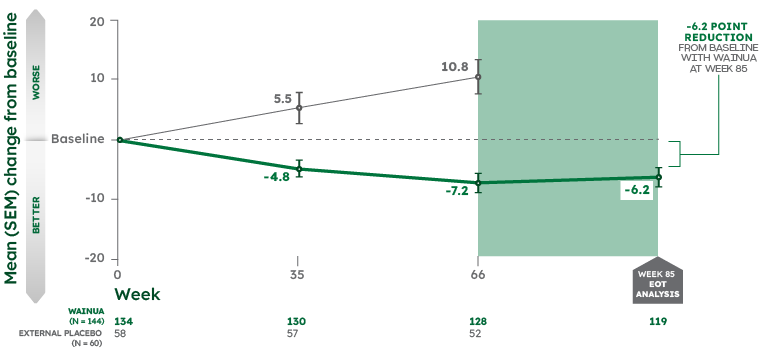
Note: From Week 66 to Week 85, there was no external placebo group because NEURO-TTR concluded at Week 66. Change in Norfolk QoL-DN total score was assessed as an exploratory outcome at Week 85. No formal statistical analyses were performed at Week 85.3
The Norfolk QoL-DN score is a validated patient-reported quality-of-life assessment for patients with hATTR-PN1,6,7
Scores measure 5 domains, including physical function/large-fiber neuropathy, activities of daily living, symptoms, small-fiber neuropathy, and autonomic nerve function.1
The higher the score, the greater the disease impact on QoL1
AUTONOMIC NEUROPATHY
Neuropathy-related impact, including orthostasis,
gastrointestinal, and genitourinary functions
PHYSICAL FUNCTION/LARGE-FIBER
NEUROPATHY
Impact of motor functions and sensory function related
to large-nerve fibers (eg, weakness, unsteadiness on
feet, unable to feel feet when walking)
ACTIVITIES OF DAILY LIVING
Impact of neuropathy on routine activities of daily life (eg, bathing, toileting, and fine finger movements)
SYMPTOMS
Impact of the common symptoms of neuropathy at 4 body sites (feet, legs, hands, and arms)
SMALL-FIBER NEUROPATHY
Impact of sensory function related to small-fibers (eg, numbness, tingling, needle-like pain, and loss of thermal sensation)
Norfolk QoL-DN includes measures for physical functioning, symptoms score, and activities of daily living1
‡Improvement is defined as a negative change from baseline; patients with missing data were counted as not improved.3
||As measured by Norfolk QoL-DN total score, validated patient-reported quality-of-life assessment for patients with hATTR-PN.1,6,7
¶The version of the Norfolk QoL-DN that was used in the trial had a range from -4 to 136 points, with higher scores representing greater impairment.1
Responder analysis for Norfolk QoL-DN total score at Week 35 and Week 661-3
Responder analysis at Week 351
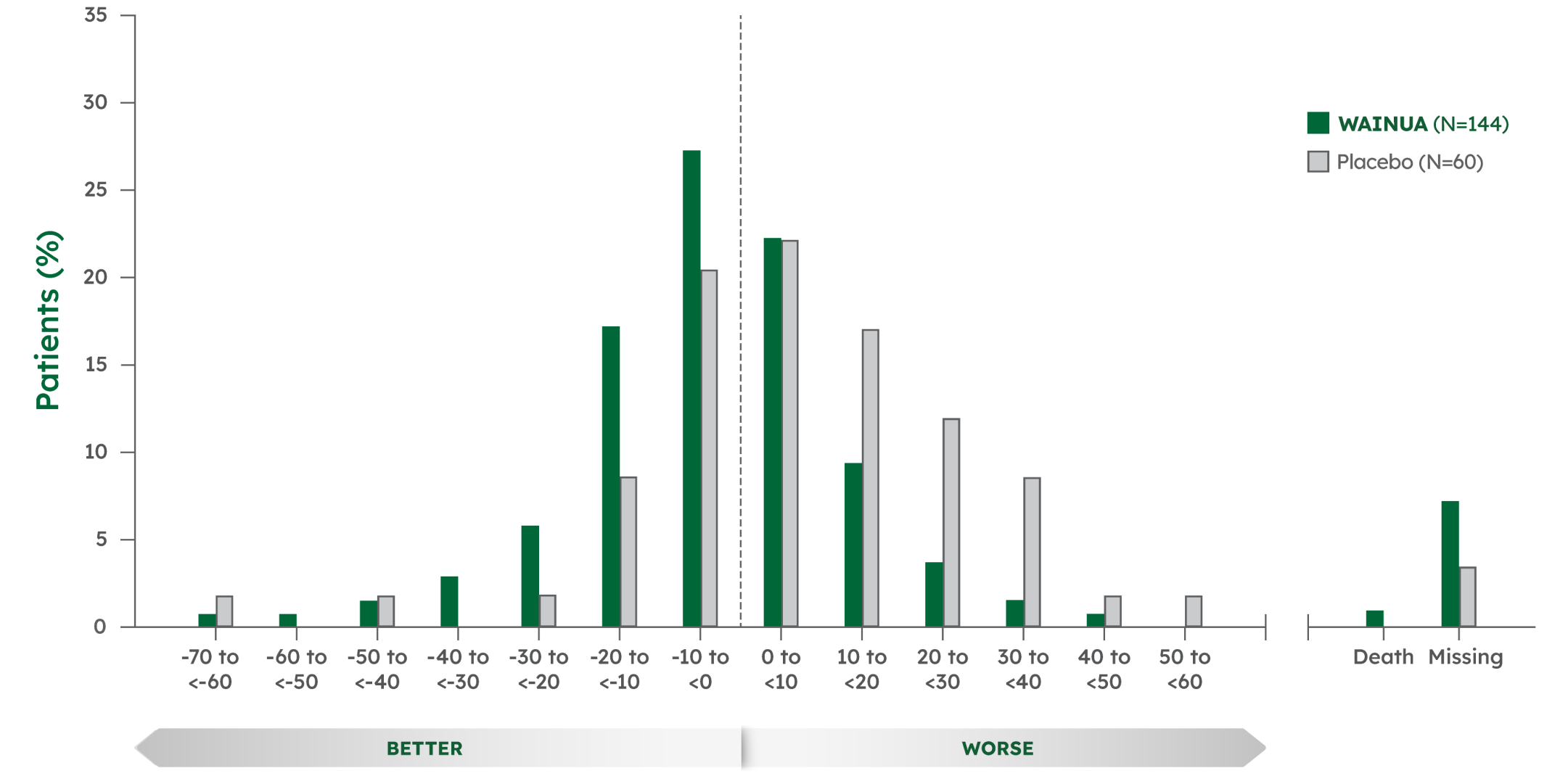
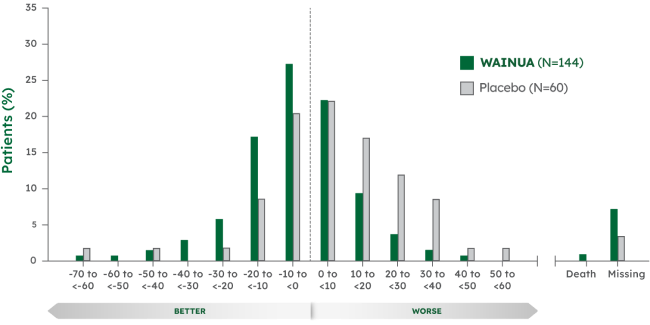
Patients treated with WAINUA had better Norfolk QoL-DN total scores vs patients treated with placebo from baseline at Week 35 and Week 66.§2,3
- Week 35: WAINUA, 56% vs placebo, 34%
- Week 66 (post hoc analysis): WAINUA, 65% vs placebo, 23%
Note: A responder (improvement) was defined as a patient whose Norfolk QoL-DN score change from baseline was less than threshold value of 0.2,3
§At Week 35, the number of patients in each group of the full analysis set was used as the denominator (WAINUA, N=140; placebo, N=59).2
At Week 66, the number of patients with nonmissing values in each group of the full analysis set was used as the denominator (WAINUA, N=128; placebo, N=52).
See the autonomic data for WAINUA
Explore the safety profile of WAINUA
FOOTNOTES
EOT, end of treatment; hATTR-PN, polyneuropathy of hereditary transthyretin-mediated amyloidosis; LSM, least-squares mean; mNIS+7, modified Neuropathy Impairment Score +7; Norfolk QoL-DN, Norfolk Quality of Life-Diabetic Neuropathy; QoL, quality of life; SEM, standard error of mean.
REFERENCES
- 1. WAINUA® (eplontersen) [prescribing information]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2025.
- 2. Data on File, REF-205487, AZPLP.
- 3. Coelho T, Marques W Jr, Dasgupta NR, et al. Eplontersen for hereditary transthyretin amyloidosis with polyneuropathy [article and supplementary online content]. JAMA. 2023;330(15):1448-1458.
- 4. Coelho T, Ando Y, Benson MD, et al. Design and rationale of the global phase 3 NEURO-TTRansform study of antisense oligonucleotide AKCEA-TTR-LRx (ION-682884-CS3) in hereditary transthyretin-mediated amyloid polyneuropathy. Neurol Ther. 2021;10(1):375-389.
- 5. Dyck PJB, González-Duarte A, Obici L, et al. Development of measures of polyneuropathy impairment in hATTR amyloidosis: From NIS to mNIS + 7. J Neurol Sci. 2019;405:1-8.
- 6. Vinik EJ, Hayes RP, Oglesby A, et al. The development and validation of the Norfolk QOL-DN, a new measure of patients' perception of the effects of diabetes and diabetic neuropathy. Diabetes Technol Ther. 2005;7(3):497–508.
- 7. Vinik EJ, Vinik AI, Paulson JF, et al. Norfolk QOL-DN: validation of a patient reported outcome measure in transthyretin familial amyloid polyneuropathy. J Peripher Nerv Syst. 2014;19(2):104–114.
IMPORTANT SAFETY INFORMATION
WARNINGS AND PRECAUTIONS
- Reduced Serum Vitamin A Levels and Recommended Supplementation WAINUA leads to a decrease in serum vitamin A levels. Supplement with recommended daily allowance of vitamin A. Refer patient to an ophthalmologist if ocular symptoms suggestive of vitamin A de ficiency occur.
ADVERSE REACTIONS
Most common adverse reactions (29% in WAINUA-treated patients) were vitamin A decreased (15%) and vomiting (9%).
INDICATION
WAINUA injection, for subcutaneous use, 45 mg is indicated for the treatment of the polyneuropathy of hereditary transthyretin-mediated amyloidosis
in adults.


